Two independent models of the ionic currents that generate the human atrial cell action potential were published in 1998, both derived from similar data but giving different action potential shapes; a spike-and-dome configuration for the Courtemanche et al. model, and a triangular action potential for the Nygren et al. model. These different action potential configurations can be seen in cells from the same preparation, so they reflect heterogeneity in atrial cell electrophysiology. Repolarisation is primarily by the delayed rectifier currents (IK,r and IK,ss) for the Courtemanche et al. model, and by the sustained K current (Isus or IK,ur) for the Nygren et al. model. Here we characterise re-entrant spiral wave solutions for isotropic two-dimensional cardiac virtual tissues constructed using these models. Spiral waves were initiated by the phase distribution method (Biktashev & Holden, 1998) in 75 mm square media for Coutemanche et al. model and 96 mm square media for Nygren et al. model with a diffusion coefficient of 0.3125 cm2 s-1 that gave a plane wave velocity of 30 cm s-1.
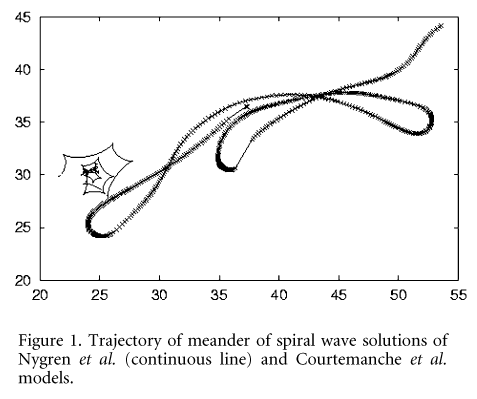
For the Courtemanche et al. model, a space step of 0.1 mm is necessary to avoid computational artifacts. With this and smaller space steps the spiral wave is stable, and shows a quasilinear meander pattern, the core following an extended 30 mm line of conduction block. Then dissipation of the excitation front in the vicinity of the spiral wave core occurs, which effectively leads to the development of a new, displaced spiral wave core, around which propagation is still possible.
For space steps of 0.2 mm and larger the spiral wave breaks down after two rotations, leading into fibrillation-like irregularity, as reported in Nygren et al. (2001). Movies show that the wave-breaking in these artifactually unstable spiral wave solutions is also produced by local dissipation of the excitation front.
For the Nygren et al. model the spiral wave is stable, even with a space step of 0.32 mm, with a period of 270 ms, and its core meanders biperiodically over 10 mm2. Both models generate stable functional re-entry consistent with atrial flutter in isotropic media.