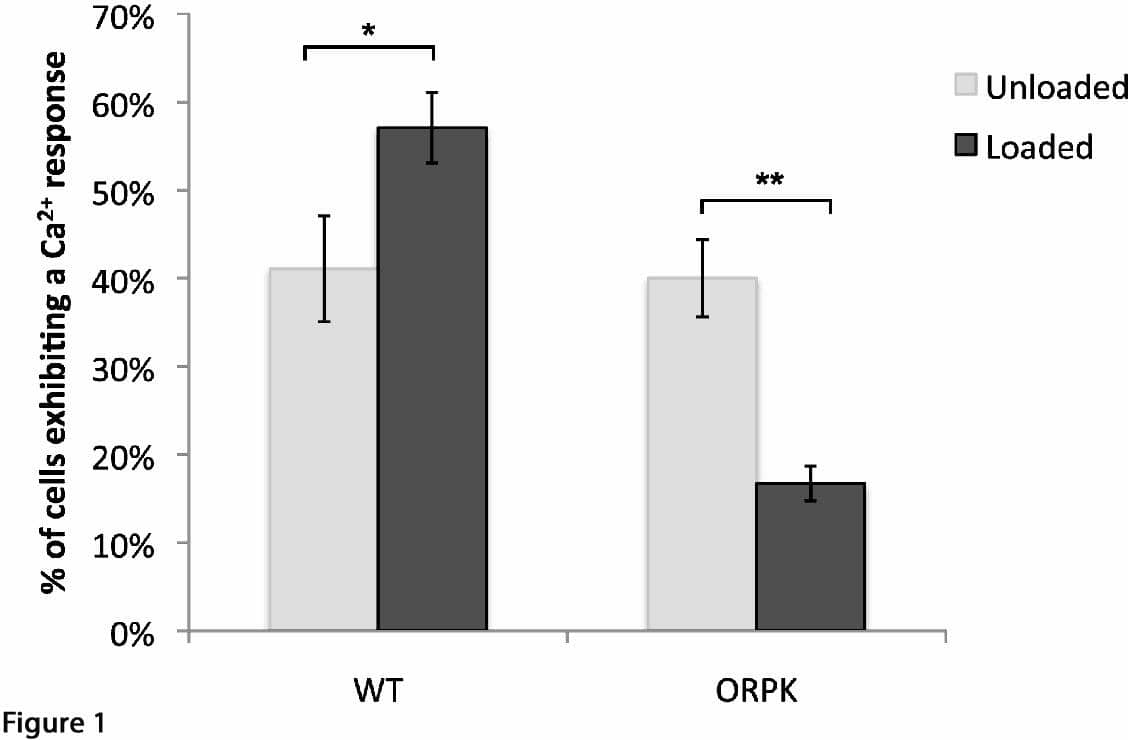
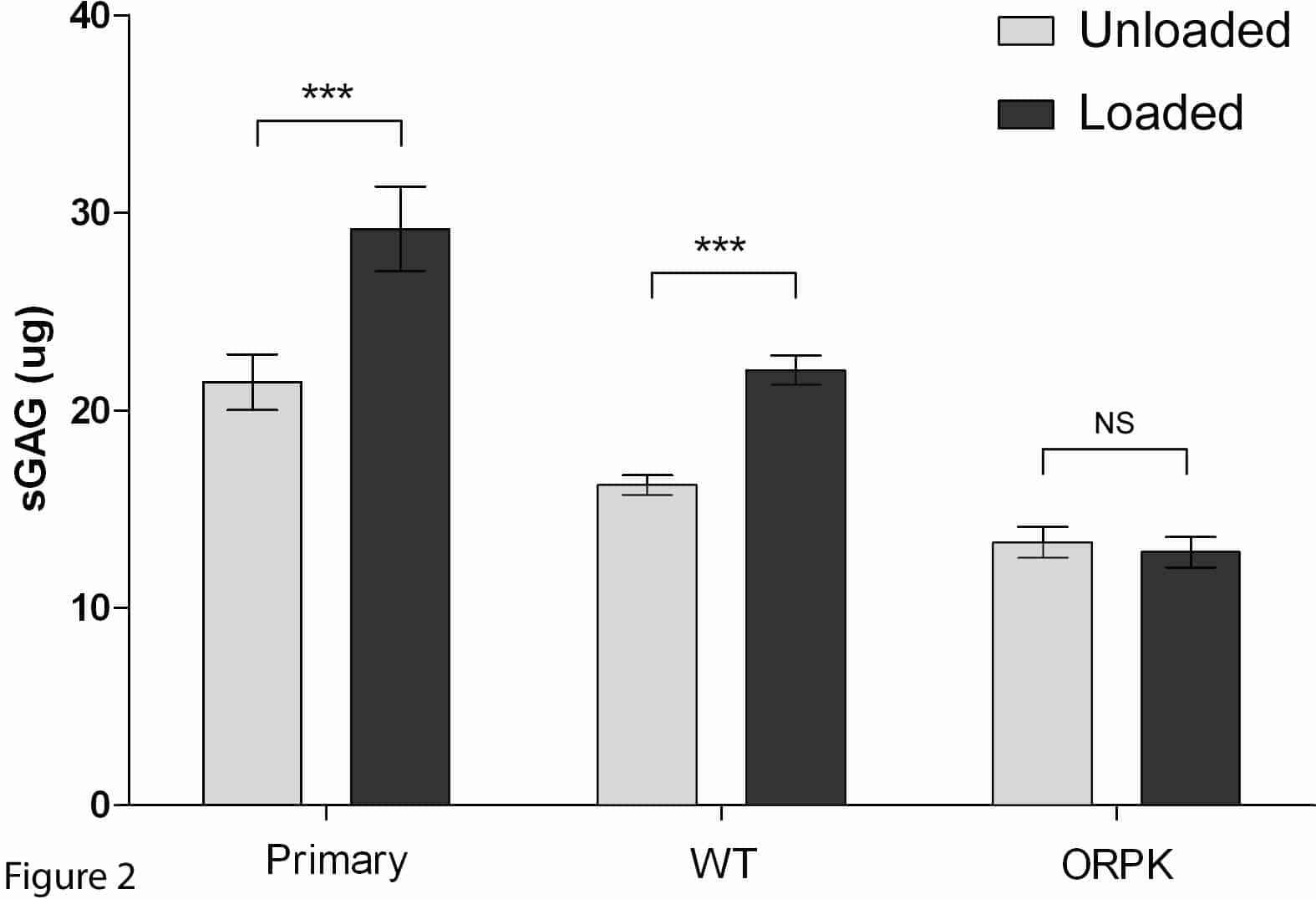
Introduction: Primary cilia are found in virtually all eukaryotic cells including chondrocytes[1]. In various cell types they serve as mechanoreceptors, linked to subsequent activation of intracellular calcium (Ca2+) signals in mechanotransduction cascades [2]. Articular chondrocytes respond to loading by increasing matrix synthesis, a response that also involves Ca2+ signalling [3]. This study tests the hypothesis that chondrocyte primary cilia are required for mechanosensitive (Ca2+) signalling and the up-regulation of sulphated glycosaminoglycan (sGAG) synthesis. Methods: Experiments used primary bovine chondrocytes and a cell line sourced from Wild-type (WT) and Tg737 Oak Ridge Polycystic Kidney (Tg737orpk) mice. Hypomorphic allele mutation of the Tg737 gene (IFT88) disrupts polaris expression, interrupting ciliogenesis [4]. WT and ORPK cells contain a temperature sensitive mutant of the SV40 large tumor antigen (T-Ag) under interferon-γ-inducible H-2Kb promoter control. Cells were expanded with 10 ng.mL-1 interferon-γ at 33°C and transformed into chondrocytes by removing interferon-γ and culturing at 37°C. Both WT and ORPK chondrocytes were seeded into 3% agarose constructs at 10 x 106 cells.mL-1. In addition, primary articular chondrocytes were isolated and seeded identically. Chondrocyte phenotype is maintained in all cell types in this model. Cilia expression was investigated by immunofluorescent labelling of acetylated α-tubulin. To investigate Ca2+ signaling, cells seeded in agarose were labeled with Fluo4, exposed to 15% static strain or left unloaded as controls and Ca2+ transients visualised by fluorescence microscopy. To investigate matrix production constructs were subjected to 24hrs cyclic compression at 1Hz, 0-15% strain or remained unloaded. Matrix synthesis was quantified by measuring sGAG using a spectrophotometric assay. Results: Immunofluorescence showed distinct, singular primary cilia in WT but an almost complete absence of cilia in ORPK cells. Static loading statistically significantly increased the number of WT cells exhibiting Ca2+ transients but conversely reduced Ca2+ signalling in ORPK cells (Fig.1). Cyclic compression induced a statistically significant increase in sGAG synthesis in primary and WT chondrocytes but the response was abolished in ORPK cells (Fig. 2). For all experiments n >24 constructs, >3 preparations. Conclusions: ORPK cells with missing or stunted cilia show markedly disrupted mechanosensitive Ca2+ signalling and mechanosensitive sGAG synthesis was abolished. Although the mechanism is unclear, these preliminary experiments provide evidence for the essential role of the primary cilium in the response of chondrocytes to loading.
University of Manchester (2010) Proc Physiol Soc 19, PC57
Poster Communications: Primary cilia are essential for chondrocyte mechanotransduction
A. K. Wann1, S. R. McGlashan2, N. Zuo2, C. J. Haycraft3, C. A. Poole4, M. M. Knight1
1. School of Engineering and Materials science, Queen Mary, University of London, London, United Kingdom. 2. Anatomy with radiology, University of Auckland, Auckland, New Zealand. 3. Department of Craniofacial Biology, Medical University of South Carolina, Charleston, South Carolina, United States. 4. Department of Medicine, University of Otago, Otago, New Zealand.
View other abstracts by:
Fig 1: Static compression increases the number of cells exhibiting Ca2+ transients in WT cells but not in ORPK cells. Values shown are means ± SEM (*/**p< 0.05’ **p<0.001, unpaired t-test)
Fig. 2: Cyclic compression up-regulates sGAG synthesis in primary and WT cells but not in ORPK cells. Values represent mean (± SEM) sGAG synthesised per construct. (***p<0.001, unpaired t-test).
Where applicable, experiments conform with Society ethical requirements.