Introduction
Inflammatory bowel disease (IBD), primarily Crohn’s disease and ulcerative colitis, is an immune-mediated inflammatory disorder of the gastrointestinal tract. Beyond intestinal pathology, individuals with IBD frequently experience extra-intestinal manifestations and have an increased risk of cerebrovascular events, potentially driven by systemic inflammation and prothrombotic mechanisms. Using a dextran sulphate sodium (DSS)-induced colitis model, we investigated whether acute intestinal inflammation is associated with systemic inflammatory changes and alterations in cerebral haemodynamics and functional connectivity assessed using functional ultrasound (fUS).
Methods
CD1 mice aged 4–8 weeks were allocated to acute DSS-induced colitis (5% w/v, 40 kDa DSS in drinking water for 3 days; n=20, 10 female) or control conditions (standard drinking water; n=20, 10 female). Disease activity index (DAI) was assessed daily using body weight change, stool consistency, and faecal occult blood testing. Following the 3-day exposure, mice underwent 15 minutes of resting-state fUS imaging (Iconeus One) under isoflurane anaesthesia. Blood was collected for plasma, and mice were perfusion-fixed with paraformaldehyde.
Faecal calprotectin and serum inflammatory/coagulation-related markers (IL-6, von Willebrand factor antigen [VWF:Ag], functional VWF activity [VWF:CBA], and ADAMTS13) were quantified using ELISA.
fUS was used to map cerebral blood volume (CBV) and functional connectivity across the brain. Twenty regions of interest (ROIs) were manually defined with reference to the Allen Mouse Brain Atlas. Baseline CBV was defined as the mean of the first 40 seconds; relative change (ΔCBV) was computed as (CBV − baseline)/baseline. ROI-wise ΔCBV time courses were extracted. Functional connectivity assessment was estimated from low-frequency (<0.1 Hz) fluctuations following detrending and confound removal. Group comparisons used t-tests, with family-wise error rate control via permutation testing (10,000 permutations); p<0.05 was considered significant. All procedures complied with the UK Animals (Scientific Procedures) Act 1986 and were approved by the University of Manchester ethical review board.
Results
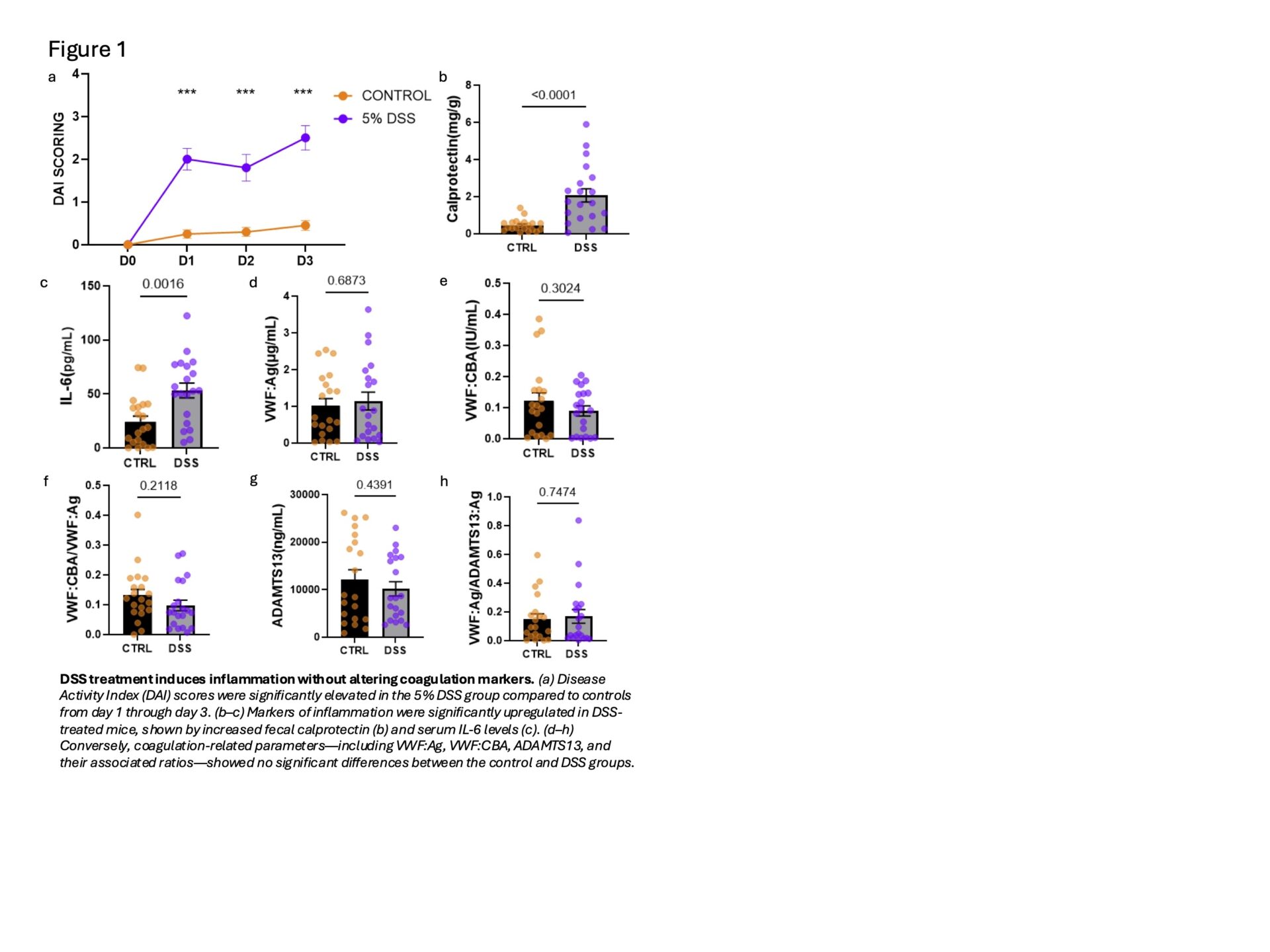
DSS exposure induced clear intestinal and systemic inflammatory changes. DAI scores increased from day 1 and remained elevated through day 3 in the DSS group relative to controls (Figure 1a). Faecal calprotectin levels were significantly higher in DSS-treated mice, alongside increased serum IL-6 (Figure 1b-c). In contrast, coagulation-related markers (VWF:Ag, VWF:CBA, and ADAMTS13) did not differ significantly between groups (Figure 1d-h).
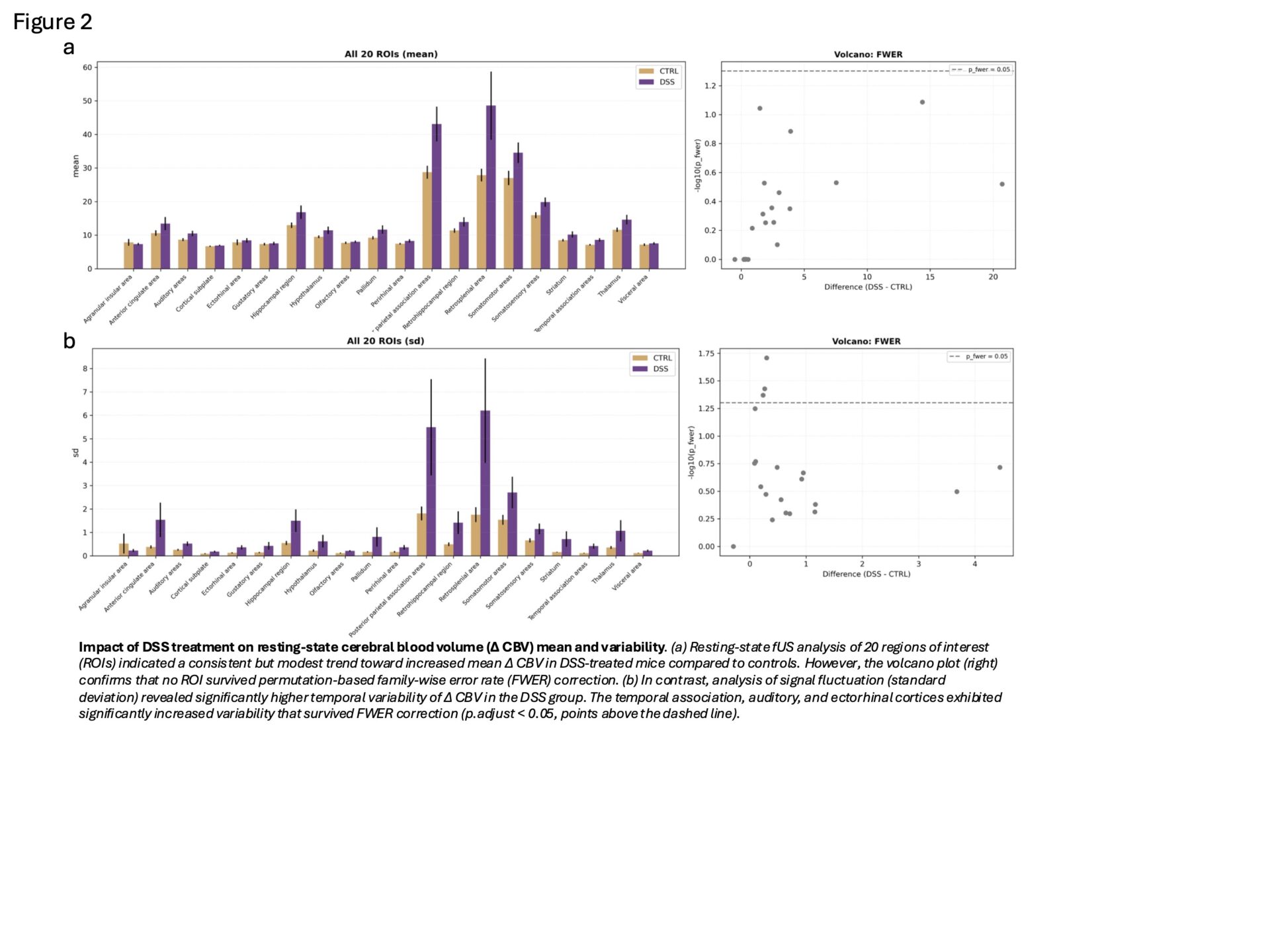
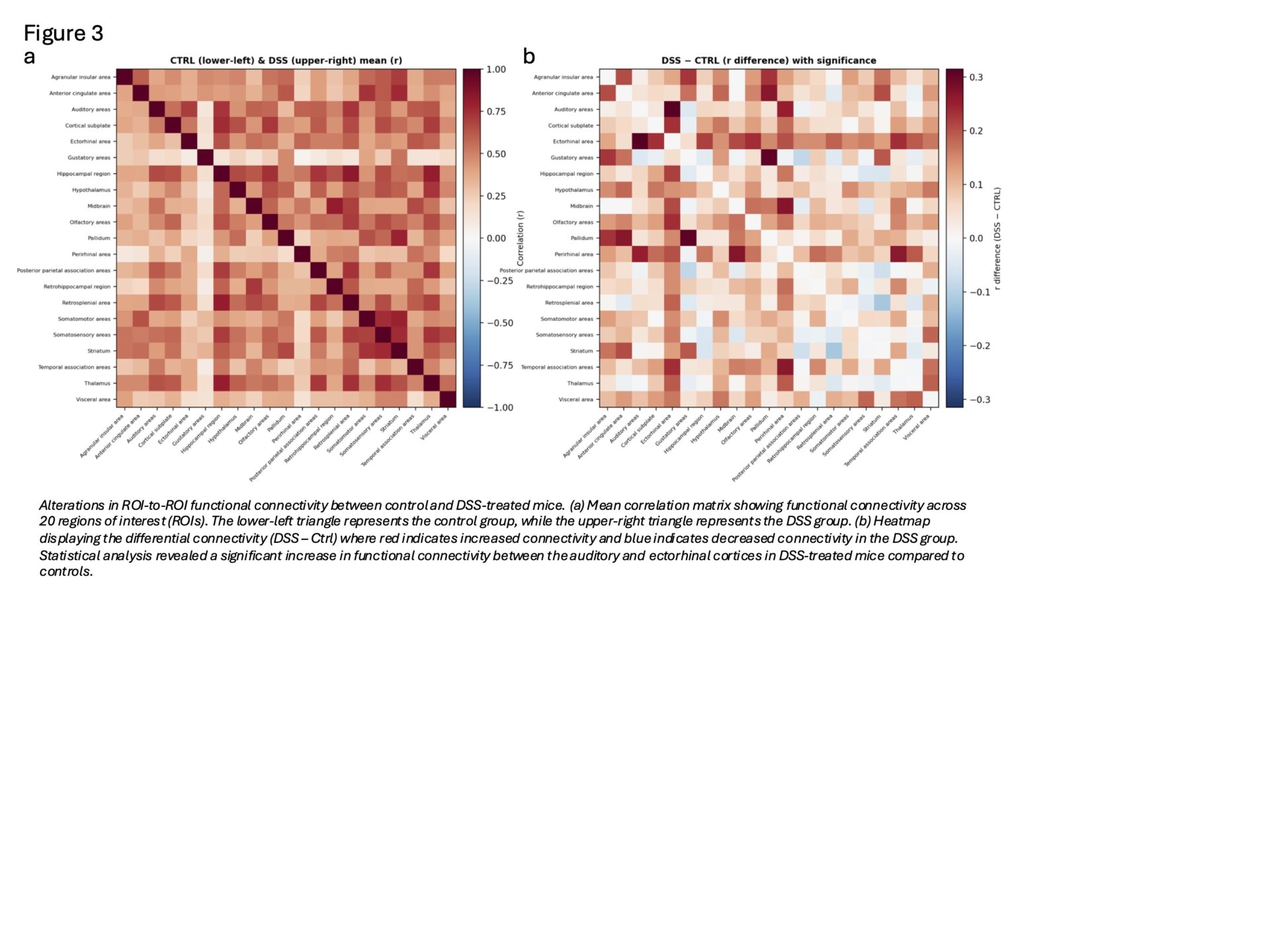
Resting-state fUS revealed a consistent but modest increase in mean ΔCBV in DSS-treated mice; however, no ROI survived permutation-based FWER correction. Notably, temporal association, auditory, and ectorhinal cortices showed significantly greater temporal variability of ΔCBV (higher standard deviation over time), suggesting increased haemodynamic fluctuation (Figure 2). ROI-to-ROI connectivity analyses further identified significantly increased functional connectivity between auditory and ectorhinal regions in the DSS group. (Figure 3)
Conclusion
Acute DSS-induced colitis produced robust intestinal inflammation accompanied by systemic cytokine elevation and measurable alterations in resting-state cerebral haemodynamic dynamics. Although mean ΔCBV differences did not survive stringent multiple-comparison correction, increased temporal variability and enhanced connectivity between auditory and ectorhinal regions suggest that peripheral inflammation may modulate neurovascular dynamics. Further mechanistic work is warranted to clarify inflammation–brain coupling in IBD and its potential relevance to cerebrovascular vulnerability.