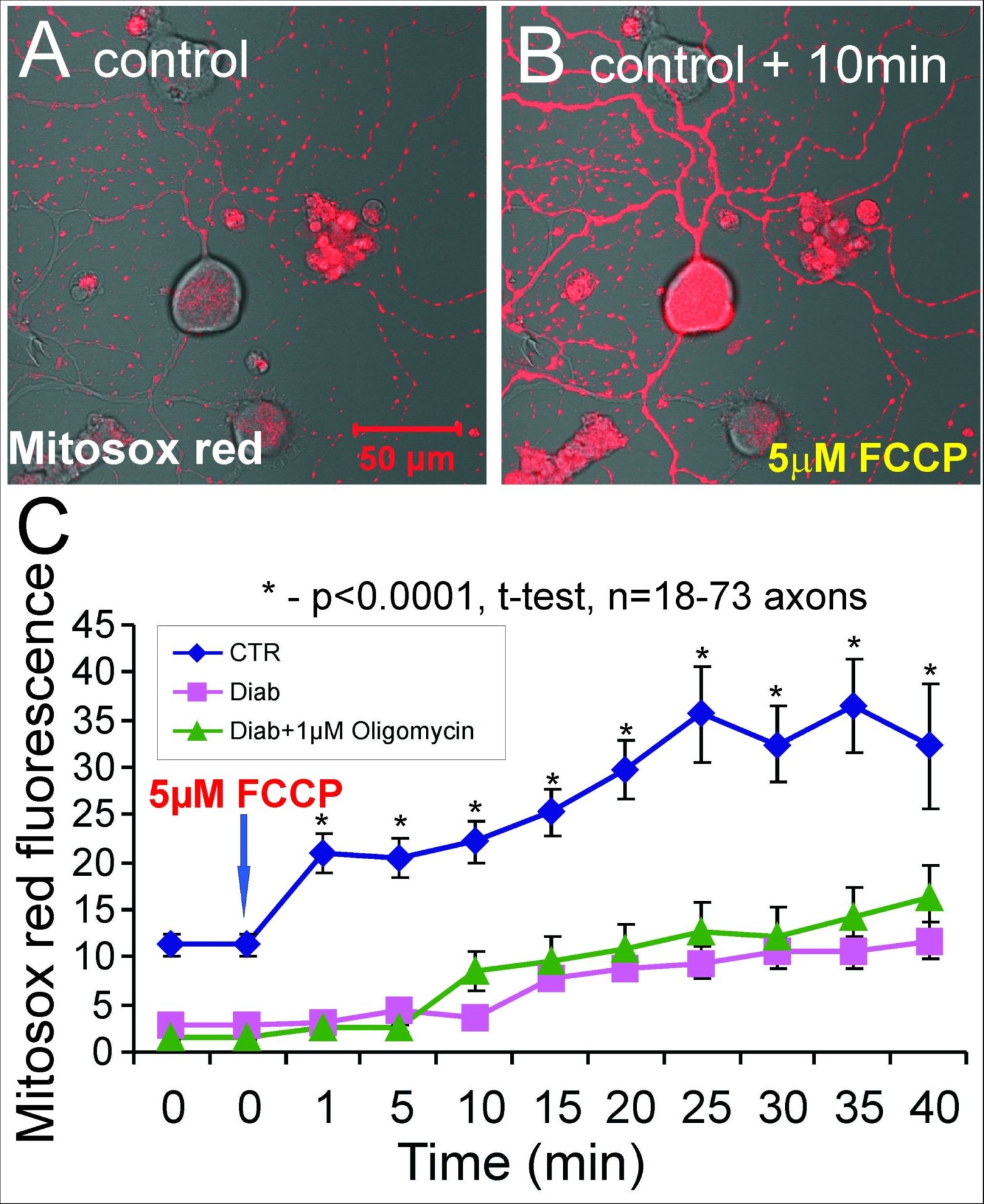
Diabetic sensory neuropathy is a major complication of type 1 and 2 diabetes that leads to distal loss of nerve fibers [1,2]. Impairments in mitochondrial function have been proposed to play a role in the etiology of the neurodegeneration, possibly through induction of oxidative stress [3,4]. We tested the hypothesis that mitochondrial dysfunction in sensory neurons in type 1 diabetes is due to abnormal activity of the respiratory chain and an altered mitochondrial proteome. Rates of oxygen consumption in mitochondria from dorsal root ganglia (DRG) of age-matched control, 12-22 week streptozotocin (STZ)-induced type 1 diabetic rats and diabetic rats treated with insulin were measured by OROBOROS oxygraph. Rates of coupled respiration with pyruvate + malate (P + M; full respiratory chain) and with ascorbate + TMPD (Asc + TMPD; Complex IV) in lumbar DRG were unchanged after 12 weeks of diabetes. By 22 weeks of diabetes, respiration with P + M was significantly decreased by 31-44% and with Asc + TMPD by 29-39% compared to control. Attenuated mitochondrial respiratory activity of STZ-diabetic rats was significantly improved by insulin treatment that did not fully correct other indices of diabetes. Enzymatic activities of mitochondrial complexes I and IV and the Krebs cycle enzyme, citrate synthase, were decreased in mitochondria from DRG of 22 week STZ-diabetic rats compared to control. Quantitative proteomic analysis using 13C6-Lys and 13C6,15N4-Arg labeled mitochondria as isotope tagged internal standards indicated that proteins associated with oxidative phosphorylation and the citric acid cycle were significantly down-regulated. Western blotting of DRG samples confirmed the proteomic analysis results for a specific subset of proteins and revealed reduced activation of AMP kinase (AMPK) coupled with diminished expression of peroxisome proliferator-activated receptor gamma coactivator-1 alpha (PGC-1α). In vitro confocal imaging studies with neurons from diabetic rats using TMRM+ in sub-quench mode showed mitochondria in axons to be depolarized and to exhibit an aberrant hyperpolarization in response to oligomycin. Parallel imaging studies using Mitosox red as an indicator of mitochondrially-generated superoxide demonstrated that axonal mitochondria of diabetic neurons had reduced levels of ROS production (see Figure). Mitochondrial dysfunction in sensory neurons in type 1 diabetes was associated with impaired rates of respiratory chain activity and modified adaption to hyperpolarization that was associated with diminished superoxide production in axonal mitochondria. The abnormal mitochondrial activity correlated with a down-regulation of an array of mitochondrial proteins and lowered activation status of the up-stream regulators of mitochondrial biogenesis, AMPK and PGC-1α.
University of Manchester (2010) Proc Physiol Soc 19, PC223
Poster Communications: Diabetes induces aberrant mitochondrial physiology and phenotype in sensory neurons: relevance to oxidative stress and associated neurodegeneration
E. Akude1,2, S. K. Roy Chowdhury1, E. Zherebitskaya1, D. R. Smith1,2, R. T. Dobrowsky3, P. Fernyhough1,2
1. St Boniface Hospital Research Centre, Winnipeg, Manitoba, Canada. 2. Pharmacology & Therapeutics, University of Manitoba, Winnipeg, Manitoba, Canada. 3. Pharmacology & Toxicology, University of Kansas, Lawrence, Kansas, United States.
View other abstracts by:
Sensory neurons from control or STZ-diabetic rats were loaded with 400nM Mitosox red - a sub-group of diabetic neurons were pre-treated for 15min with oligomycin to hyperpolarize the mitochondria and maximize take up of the dye. FCCP was added to uncouple the mitchondria and induce maximal rates of respiratory chain electron transfer and hence increase electron leakage and ROS production. The data in C shows that mitochondria in axons of neurons from diabetic rats have a reduced capacity to generate ROS derived from the respiratory chain.
Where applicable, experiments conform with Society ethical requirements.