Introduction: It is well established that adipose tissue has a strong influence on whole-body insulin sensitivity, glucose and lipid metabolism. Previous studies examining mitochondrial function in muscle in obese individuals have shown lower expression of transcript levels of genes related to oxidative phosphorylation and lower transcription factors (e.g., PGC-1α) suggesting decreased mitochondrial biogenesis. Although research interest related to adipose tissue bioenergetics in overweight and obese individuals has been increasing in recent years, only one study, to our knowledge, has examined how human adipose tissue correlates to a ketogenic diet. However, this study had only 5 participants in each condition, and diet was self-reported. Therefore, to our knowledge, this will be the first study to explore adipose mitochondrial bioenergetics in humans, and how it responds to a 4-week intervention following a ketogenic diet or ingesting a ketone ester.
Aims/ Objectives: The aim of this exploratory analysis is to a) understand how a ketogenic diet or ketone supplementation influences mitochondrial respiratory capacity in the adipose tissue, and b) if there are correlations between baseline values of mitochondrial respiration in the adipose tissue and other baseline measurements.
Methods: Currently, 33 individuals (age 45±12 years; 24 females, BMI 30.0±3.6) have been randomised to 4 weeks of either a control (n = 12, habitual diet), ketone supplementation (n = 11, thrice daily alongside habitual diet) or ketogenic diet (n = 10) condition (all prescribed at energy balance), with 23 having completed their respective interventions. Baseline measurements (e.g., body mass, body composition via dual energy x-ray absorptiometry, PAEE) and mitochondrial respiration in the adipose tissue were assessed before and after the intervention. Mitochondrial bioenergetics were established by measuring through high-resolution respirometry (Oxygraph-2k, Oroboros Instruments, Innsbruck, Austria) by following a substrate-uncoupler-inhibitor titration (SUIT) protocol SUIT-008 DL (DIG-1PM-2D-2c-3G-4S-5U-6Rot-7Ama). The protocol consisted of adding specific substrates related to each mitochondrial complex (e.g., succinate for Complex II) to investigate its contribution to the electron transport chain (ETC). Change from baseline to follow-up were compared between groups with a one-way ANOVA.
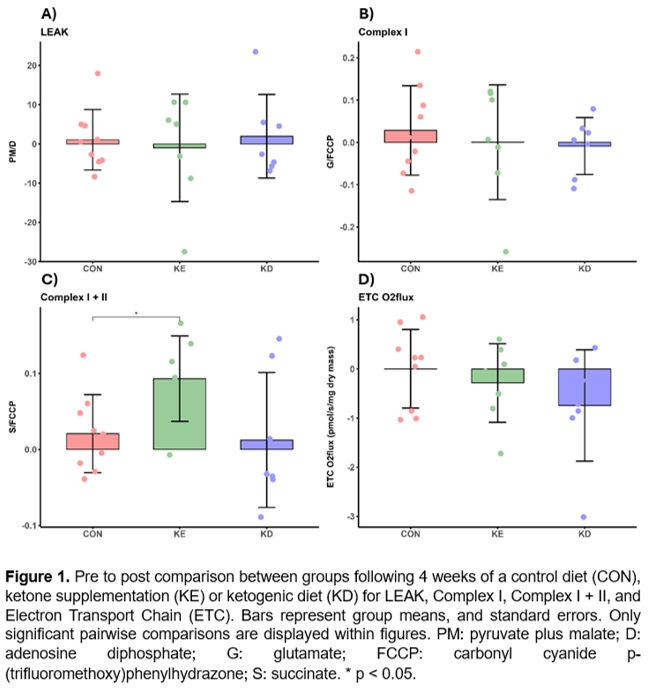
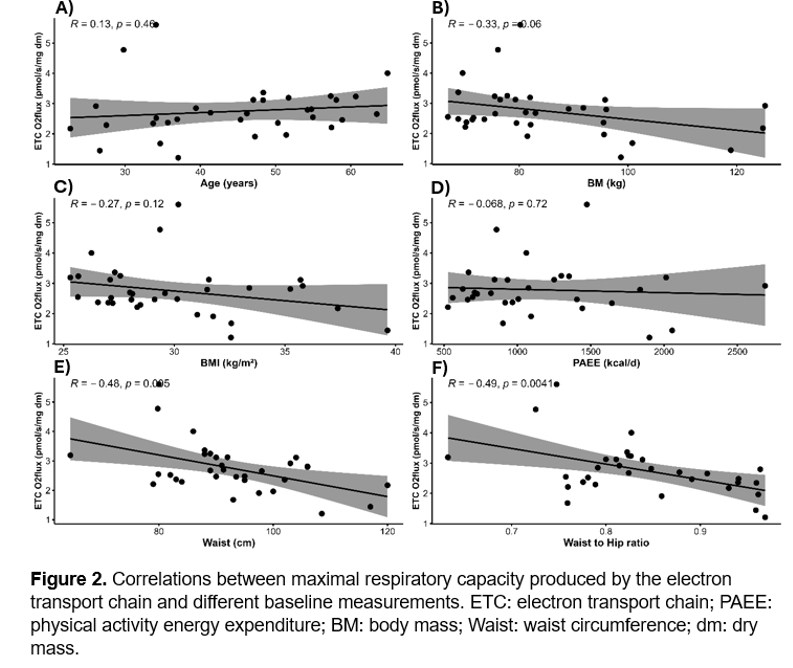
Results: There were no significant differences between groups in relation to LEAK (p > 0.05, Fig-1A), Complex I (p > 0.05, Fig-1B) and ETC (p > 0.05, Fig-1D). However, there was a significant difference in Complex I+II linked respiration between the control group and ketone supplementation group (p < 0.05, Fig-1C). Significant negative correlation was found between the total release of energy by the electron transport chain in the adipose tissue and waist circumference (R = -0.48, p = 0.005, Fig-2E) and waist to hip ratio (R = -0.49, p = 0.004, Fig-2F). No other significant correlations were found among baseline measurements.
Conclusion: Ingestion of ketone supplementation significantly increases the activity of mitochondria’s complexes I and II in the adipose tissue in relation to a standard diet. Possibly via direct contribution of the ketone ester oxidation to NADH and acetoacetyl-CoA for the mitochondrial respiration. Additionally, correlation results indicate that higher waist circumference and waist-to-hip ratio are associated with lower baseline oxidative phosphorylation capacity.