Introduction: The circadian timing system, metabolism and nutrition are closely interlinked (1). Previous research from our group has shown that, in conditions of energy balance, a 5-hour delay in meal timing causes a delay in plasma glucose phase, while melatonin, a marker of the central clock, remains aligned to the light-dark cycle (2,3). We hypothesised that during negative energy balance, central clock markers would exhibit a phase shift following a 5-hour delay in meal timing.
Methods: Healthy male participants were recruited to allow comparison with previous work (2). For 10 days leading up to the laboratory session, participants maintained a consistent sleep-wake cycle and meal timing. Participants then entered the laboratory for a 13-day/night residential protocol, during which their environment and behaviours, including dietary intake, were strictly controlled. The first two days in the laboratory were spent in energy balance, with 3 meals provided 0.5, 5.5, and 10.5 hours after waking, respectively. Participants then underwent a “constant routine” [CR1], a 37-hour protocol which enables measurement of endogenous circadian rhythms, followed by six consecutive days with a 30% energy deficit and meal timing delayed by 5 hours. Participants then underwent a second “constant routine” [CR2] to measure circadian rhythms, which were compared to CR1.
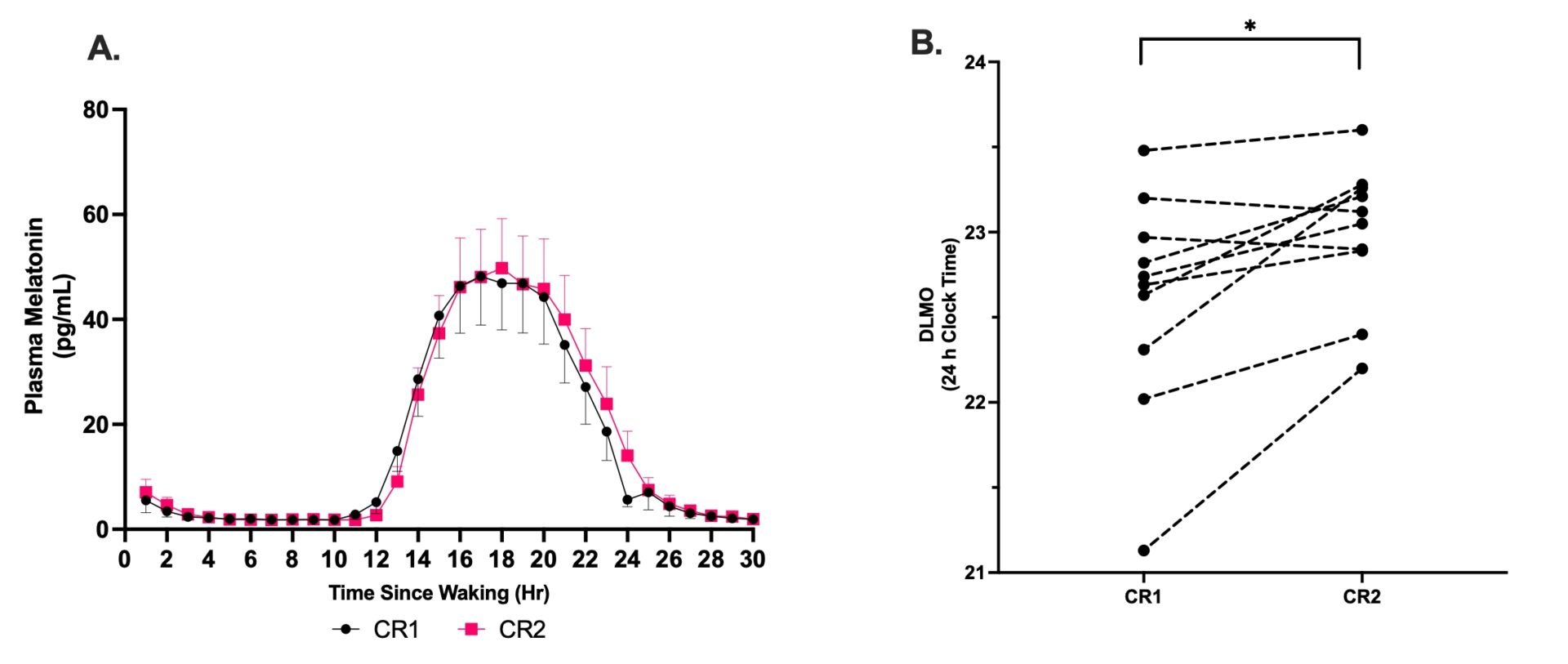
Results: Ten participants (age = 30 ± 4.4 years; BMI = 25.4 ± 1.0 kg/m2; mean ± SD) were included in the final analysis. Mean energy intake during the energy balance control days was 2,371kcal/d [± 186], and mean energy intake during the energy deficit days was 1,693kcal/d [± 144]. Mean weight loss during the energy deficit was –1.3% [± 0.8%]. There was a significant phase delay in dim-light melatonin onset [DLMO] from CR1 to CR2 [CR1 = 22:36 h ± 00:39 h; CR2 = 23:00 h ± 00:24 h; difference = 24mins; 95% CI, 6–41min; p = 0.013; paired t-test]. The plasma cortisol acrophase was also significantly delayed between CR1 [23:58 h ± 00:15 h] and CR2 [24:35 h ± 00:17 h] by 36 minutes [95% CI, 20–52 mins; p = 0.0001; 2-sample summary t-test]. There was a significant delay in plasma glucose acrophase between CR1 [13:52 h ± 01:21 h] and CR2 [20:39 h ± 00:54 h] by 06:46 h [95% CI, 05:40 h to 07:52 h; p < 0.0001; 2-sample summary t-test]. Finally, there was a significant delay in the acrophase of HDL-C from CR1 to CR2 [CR1 = 10:57 h ± 00:53 h; CR2 = 12:11 h ± 00:30 h; difference = 01:14 h; 95% CI, 00:32 h to 01:56 h; p = 0.001; 2-sample summary t-test]. There was no significant phase shift in triglycerides, total or LDL-cholesterol.
Conclusions: The phase delay in plasma glucose rhythms is consistent with previous work demonstrating meal timing as a dominant time-cue for the circadian control of glucose. However, our findings demonstrate for the first time a delay in markers of human central clock phase. This reveals an increased potential of chrono-nutrition interventions to regulate the human circadian system.