By Tashi Raina and Alireza Mani from University College London (UCL), UK
The Historic Objects and Collections at UCL series presents Physiological Instrument #028: a Haldane apparatus on display in the A.V. Hill Lecture Theatre on UCL’s main campus in Bloomsbury, London. In this series, the team shares the stories of physiological artefacts housed within the Institute’s rich collection.
The third object featured is a Haldane apparatus, a chemical gas-analysis instrument used to analyse air composition. This version of the device at UCL dates from around 1923–1924 and was used by A.V. Hill when he took up the post of Jodrell Professor of Physiology at UCL.
From mining disasters to gas analysis
The late 19th century saw the glory of the Industrial Revolution, responsible for the creation of steam engines, factories for mass production of goods, railways and urban heating. Coal was the life blood for this new era of living, which forced mining to much larger and more drastic scales. Mines increased in depth and complexity with vast underground networks with dangerous conditions and restricted ventilation. Coal mining became one of the deadliest occupations. Hundreds of miners died every year in large disasters and many more died from lung disease or gas exposure.
A notable incident was the Tylorstown colliery explosion in 1896; where 57 miners (men and boys) died. John Scott Haldane, a Lecturer in Physiology at the University of Oxford, was commissioned by the British Home Office to investigate mining incidents. Haldane studied explosions at Tylorstown, Brancepeth and Micklesfield. His examination revealed that most miners were alive after the initial blast and their blood was found to be bright red, a hallmark of carbon monoxide poisoning. This insight demonstrated that the toxic after-damp gas was the real killer (1); and once Haldane discovered the dangers these gases presented, he needed a way to measure the concentrations accurately. This gave birth to the development of the Haldane apparatus (2).
The Haldane gas analysis apparatus
The Haldane apparatus (Figure 1) is a gas analysis device used to measure the concentrations of gases in air samples. Before its invention, gas composition was difficult to measure with accuracy as there was no reliable method to measure remaining oxygen (O2) levels or the accumulation of carbon dioxide (CO2) or carbon monoxide (CO). After-damp is the toxic mixture of gases left after an explosion; composed of CO2, nitrogen, reduced O2, and CO which is the main killer. Carbon monoxide binds to haemoglobin with 200 times the affinity of oxygen; forming carboxyhaemoglobin, which prevents oxygen transport.
Miners often remained conscious after the initial explosion, then attempted to escape and collapsed. This being the reason why the bodies were found with no obvious injury. This had confused people for decades: lamps still burnt in after-damp air indicating that oxygen was present, and yet miners and rescuers who entered post-explosion died. Carbon monoxide does not extinguish a flame, therefore flame tests were useless, and why Haldane’s gas analysis was so crucial.
John Scott Haldane and carbon monoxide poisoning
John Scott Haldane (1860-1936) was a Scottish physiologist and a pioneer in respiratory physiology. He laid foundations for future environmental physiology study. Haldane studied medicine at the University of Edinburgh and then worked at the University of Jena in Germany where he adopted an experimental approach towards physiology.
Haldane famously used himself as the test subject for his hypoxia studies, carbon monoxide exposure, as well as for the testing of the Haldane apparatus. Haldane was chosen to investigate the Tylorstown disaster as he was well established as a respiratory physiologist and could bridge the gap required between academia and field investigation. He recognised that the cause of death was contested due to uncertainty of air composition and developed a mechanism of measuring it.
Haldane was responsible for the use of canaries as a widespread practice within the mines. Before his research, miners were dependent on flames in lamps, smells, a feel of dizziness or headache. There was no real protocol; let alone an understanding of why smaller animals such as rodents would be increasingly sensitive to carbon monoxide. Haldane’s research showed that smaller animals would show symptoms of noxious gas exposure earlier due to their high metabolic rates and efficient lungs.
Canaries could show rapid behavioural changes of distress that acted as a buffer of time for miners to escape to safe levels of oxygen. After Haldane’s research, canaries became official equipment for all miners. They were endorsed by mine safety authorities, as well as the Home Office, and saved countless lives.
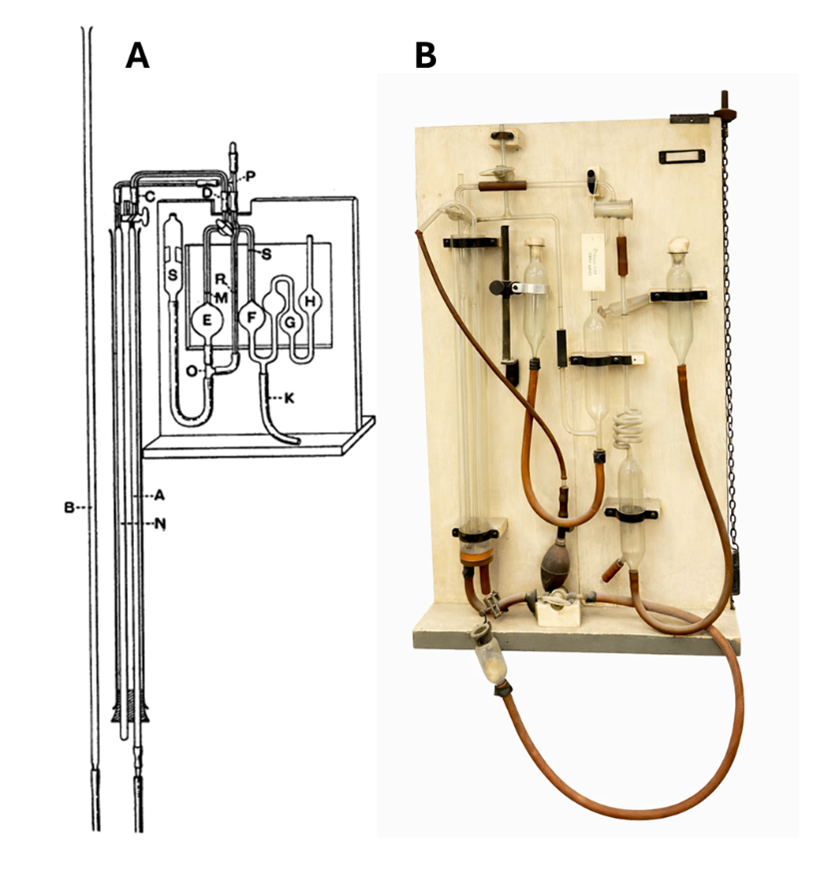
Figure 1. A. Schematic illustration of the gas analysis apparatus developed by J. S. Haldane. The diagram is taken from Haldane’s 1898 paper published in The Journal of Physiology (2).
B. The Haldane apparatus on display in the A. V. Hill Lecture Theatre at UCL, dating to approximately 1923-1924.
How does the apparatus work?
The apparatus functions by determining the percentage composition of gases within the air through selectively absorbing gases and measuring the change in volume under constant pressure and temperature. A fixed volume of air is drawn into a calibrated glass burette; this starting volume is the reference point. The air sample is passed into an absorption chamber containing potassium hydroxide which reacts with carbon dioxide and forms dissolved potassium carbonate. The remainder of the air (oxygen, nitrogen and other gases) returns to the burette and the volume drops.
The volume decrease is equal to the volume of carbon dioxide absorbed and is expressed as a percentage of the original air sample. The remaining air is then passed through a second chamber containing alkaline pyrogallol, which selectively absorbs oxygen and forms a brown oxidation product. The removal of oxygen from the gas phase causes a further reduction in volume, allowing the oxygen content to be determined.
Similarly, carbon monoxide is measured by passing the remaining gas into another chamber containing ammoniacal cuprous chloride solution, which selectively absorbs carbon monoxide by forming a chemical complex. The gas is then returned to the burette, and the further reduction in volume is measured to determine the amount of carbon monoxide present. Nitrogen (N₂) remains unabsorbed; therefore, the residual gas is assumed to consist predominantly of nitrogen. Each gas is absorbed separately to prevent overlap and ensure accurate measurement.
Physiology shaping public policy
In 1906, Haldane designed a more convenient, portable form of gas analysis apparatus that could be used in the field (3). After explosions took place, air samples were collated and the air composition was measured through the Haldane apparatus. This was to determine whether it was safe to re-enter, why the miners had died, and what the state of ventilation was. This saw a reduction in secondary waves of deaths among rescue workers.
The apparatus was used to compile evidence in official reports, set safe limits for concentrations of gases and influence mine design and protocol; in what is considered one of the earliest examples of physiology shaping public policy (4). The same principles and methods that Haldane applied to his invention were further incorporated into submarine air monitoring and tunnel development, as well as aviation and construction.
Whether it is a medical or engineering setting, the Haldane apparatus is the ancestor of all modern-day devices that are relied upon to measure gases. Although today it has been replaced by more cutting-edge technology, it was born out of necessity; and its legacy remains to be the innumerable lives saved in those mines.
Haldane and interdisciplinary approach to science and public health
Haldane was one of the clearest early examples of an interdisciplinary physiologist. His work consistently bridged laboratory science, clinical medicine, engineering, and public policy.
In 1908, he delivered an address to the Physiological Section of the British Association for the Advancement of Science entitled The Relation of Physiology to Physics and Chemistry, in which he emphasised the integration of physiology with the physical sciences (5). In addition to developing the Haldane gas analysis apparatus, he invented and designed a range of practical devices, including respirators for mine rescue, decompression protocols for divers (6), and early respiratory equipment for oxygen therapy in clinical practice (7).
Haldane also confronted some of the major crises of the early twentieth century, including the First World War (8) and the 1918 influenza pandemic (9). His response was marked by innovation: he developed methods to identify poisonous gases, such as chlorine used in chemical warfare, and contributed to the design of the “Black Veil” respirator, an early military gas mask intended to prevent gas poisoning (8).
His interdisciplinary approach extended beyond drawing on chemistry, physics, and physiology. It also incorporated a societal dimension. He worked extensively on environmental public health and co-founded The Journal of Hygiene in 1901 (now Epidemiology & Infection journal), which became one of the first major journals to publish systematic research on population health and preventive medicine. The Haldane interdisciplinary approach was continued by many of his fellow scientists, including his own son, JBS Haldane (Professor of Genetics and later of Biometry at UCL), a polymath who made original interdisciplinary contributions to physiology, genetics, evolutionary biology, and mathematics.
If you missed the first two artefacts on display by the UCL Historic Objects and Collections team, read their blogs, The Kymograph and Bárány’s Box, to discover more about the history of the inventors and their physiological instruments.
References:
1.Haldane JS. (1896). Report to the Secretary of State for the Home Department on the causes of death in colliery explosions and underground fires, with special reference to the explosions at Tylorstown, Brancepeth and Micklefield. London: HMSO (Eyre and Spottiswoode).
2.Haldane JS. (1898) Some Improved Methods of Gas Analysis. The Journal of Physiology. 22(6):465-80. doi: 10.1113/jphysiol.1898.sp000707.
3.Haldane JS. (1906) A Convenient Form of Gas Analysis Apparatus. The Journal of Hygiene. 1906 Jan;6(1):74-6. doi: 10.1017/s0022172400002667. PMID: 20474246; PMCID: PMC2236044.
4.Carter T. (2004). British Occupational Hygiene Practice 1720–1920. The Annals of Occupational Hygiene, 48(4), 299–307.
5.Haldane JS. (1908) An address on the relation of physiology to physics and chemistry: Delivered before the Physiological Section of the British Association for the Advancement of Science, Dublin, 1908. British Medical Journal. 2(2489):693-6. doi: 10.1136/bmj.2.2489.693.
6.Boycott A E., Damant, G. S., & Haldane, J. S. (1908). The prevention of compressed-air illness. The Journal of Hygiene, 8(3), 342-443. doi: 10.1017/s0022172400003399.
7.Haldane, J.S. (1929) The therapeutic administration of oxygen. British Medical Journal. 1917 Feb 10;1(2928):181-3. doi: 10.1136/bmj.1.2928.181.
8.Beckett I., Bowman T., & Connelly M. (2017). “The Western Front, 1915.” In The British Army and the First World War. Cambridge University Press.
9.Haldane JS. (1929) The influenza epidemic of 1918: colour of the blood in fatal cases. British Medical Journal 1(3571):1070. doi: 10.1136/bmj.1.3571.1070.

