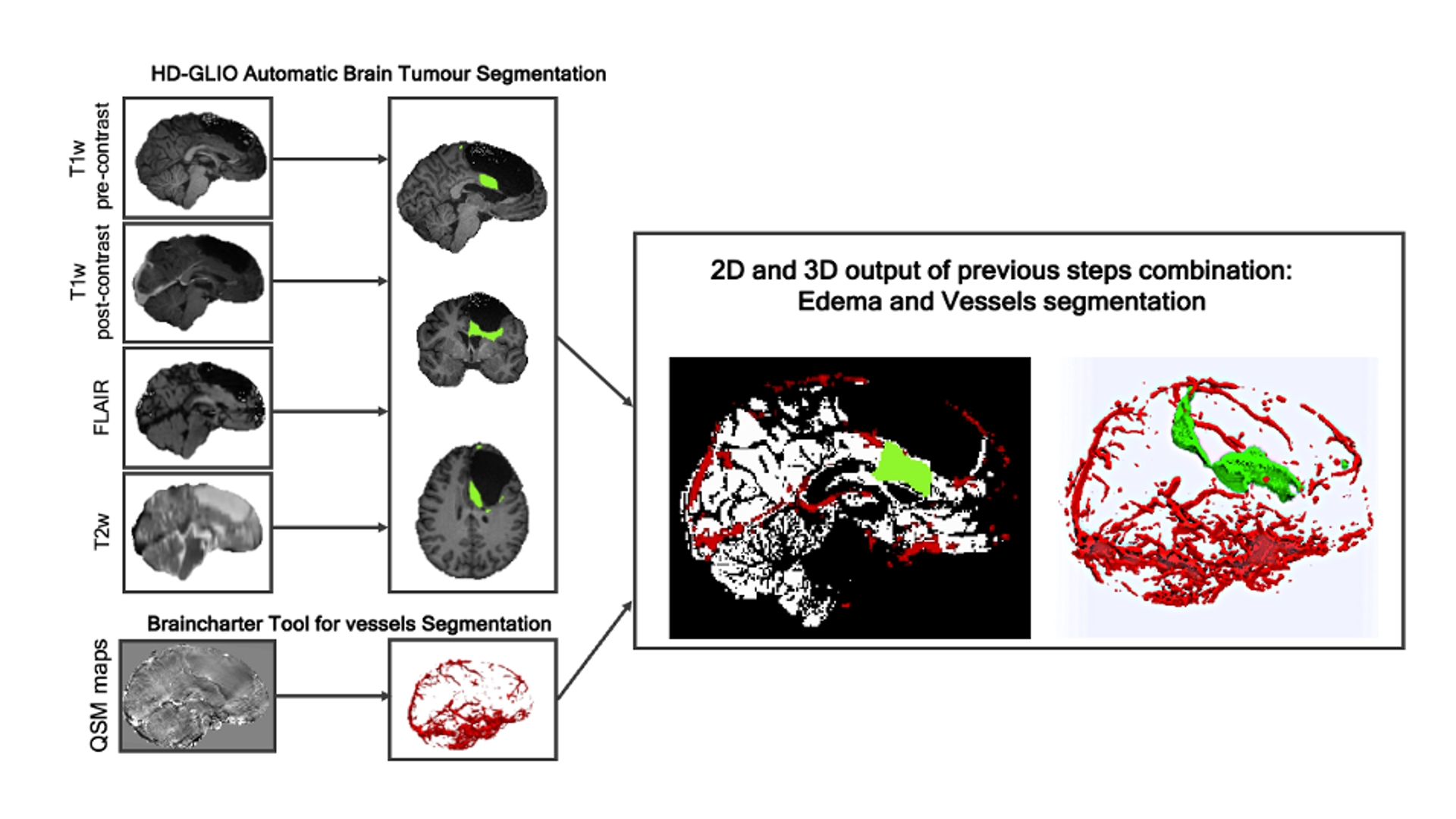
INTRODUCTION
Gliomas generate an additional blood supply to meet their increased need for nutrients and oxygen, altering normal physiology and tissue metabolism1,2. As proliferating cells demand more blood flow, the structure and function of the brain’s vasculature are disrupted. Vascular proliferation is a pathological hallmark of gliomas, leading to abnormal and dysfunctional vessels3. Considerable heterogeneity exists within tumours regarding vascular morphology. For example, high microvessel density is characteristic of astrocytic gliomas, and high-grade tumours exhibit pronounced neovascularization4.
The formation of new blood vessels to support tumour growth has clinical consequences, most notably the development of vasogenic edema, this can lead to an increase in intracranial pressure due to leakage of the blood–brain barrier5.
AIM
This study used advanced MRI techniques to characterize vascular differences across glioma subtypes.
METHODS
The study was approved by the NHS Research Ethics Committee, Wales, UK and Cardiff University’s Ethics Committee, School of Psychology, Cardiff, UK.
Quantitative susceptibility maps (QSM) were acquired from 27 patients (mean age 45.5 ± 8.8 years; 20 male) diagnosed with glioma. Vascular parameters—including vessel density, vessel diameter, and oxygen extraction fraction (OEF)—were extracted from the QSM maps.
Pre- and post-contrast T1-weighted images (gadolinium-enhanced), T2-weighted images, and T2-FLAIR were used to segment brain tissue into tumour core (enhancing vs. non-enhancing), edema, necrotic area, cavity, and residual healthy grey matter. (Figure 1)
ANOVAs were conducted to evaluate differences in OEF (within skeletonized vessels), vessel diameter, and vascular density in edema, tumour core, and residual healthy grey matter. Gender was included as a covariate.
RESULTS
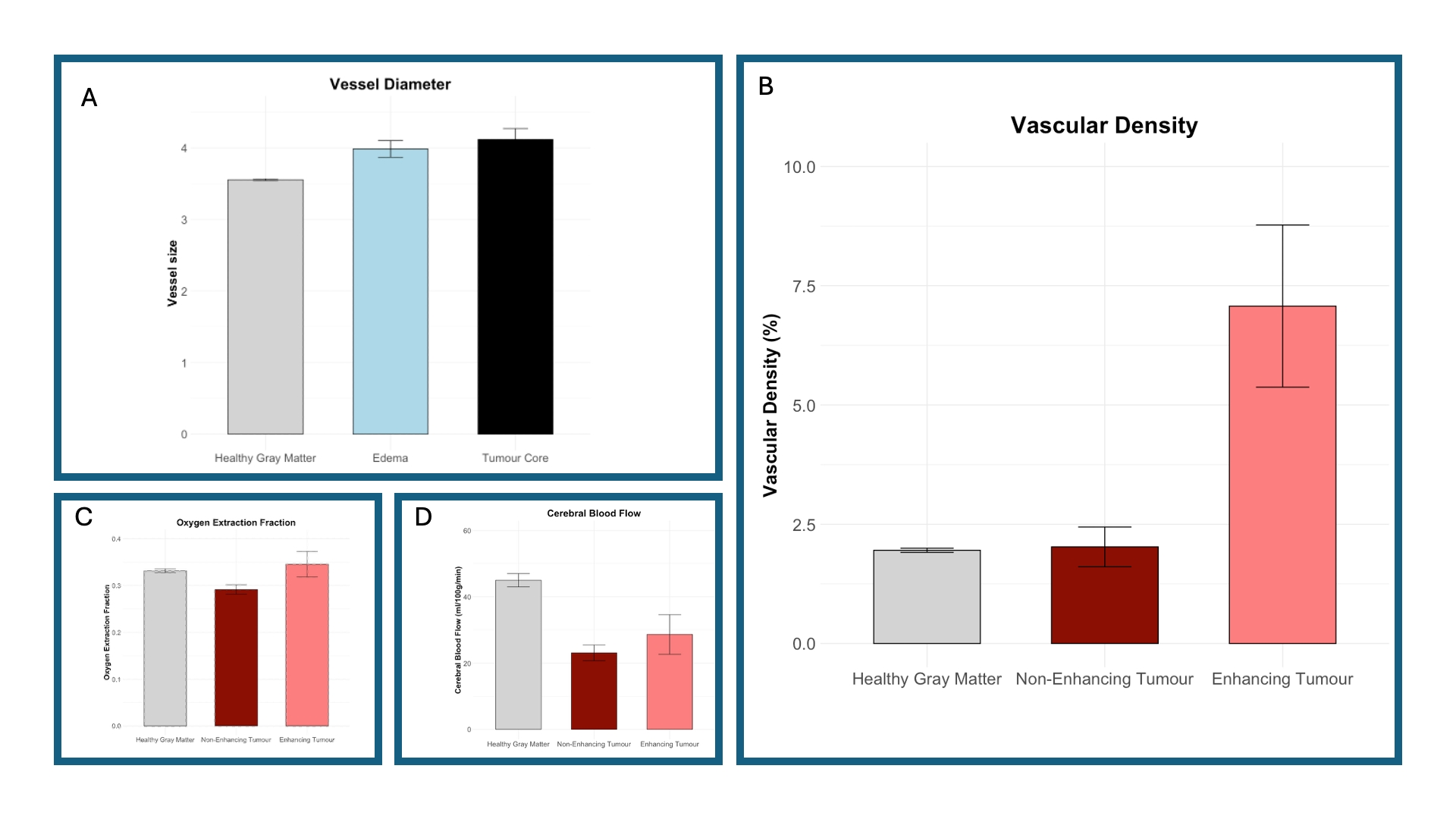
Structurally, we observed increased vessel diameter in edema and tumour core regions compared with residual healthy grey matter (F(2,38) = 11.08; p < 0.01) (Figure 2-A). Vascular density was higher in enhancing tumour tissue compared with non-enhancing tumour regions and healthy grey matter (F(2,38) = 16.81; p < 0.01). (Figure 2-B)
Physiologically, OEF was reduced in non-enhancing tumour regions compared with healthy grey matter and enhancing tumour tissue. This result may reflect infiltration or hypoxia, with altered OEF and modest vascular changes consistent with metabolic stress or tissue loss (F(2,38) = 7.16; p < 0.01) (Figure 2-C).
Cerebral blood flow (CBF) was reduced in both enhancing and non-enhancing tumour areas compared with healthy grey matter, indicating vascular–metabolic decoupling in enhancing cores likely related to abnormal vessel structure and shunting (F(2,38) = 22.07; p < 0.01) (Figure 2-D).
CONCLUSION
Our findings demonstrate the potential of QSM to identify distinct metabolic and vascular characteristics across glioma subtypes, consistent with evidence that more aggressive tumours develop their own blood supply, leading to altered vascular parameters.
Integrating QSM into routine glioma assessment may enable non-invasive hypoxia evaluation, differentiation from non-tumour abnormalities, and support individualized treatment planning. This approach could help guide surgical and radiotherapy strategies and facilitate longitudinal monitoring of treatment response.