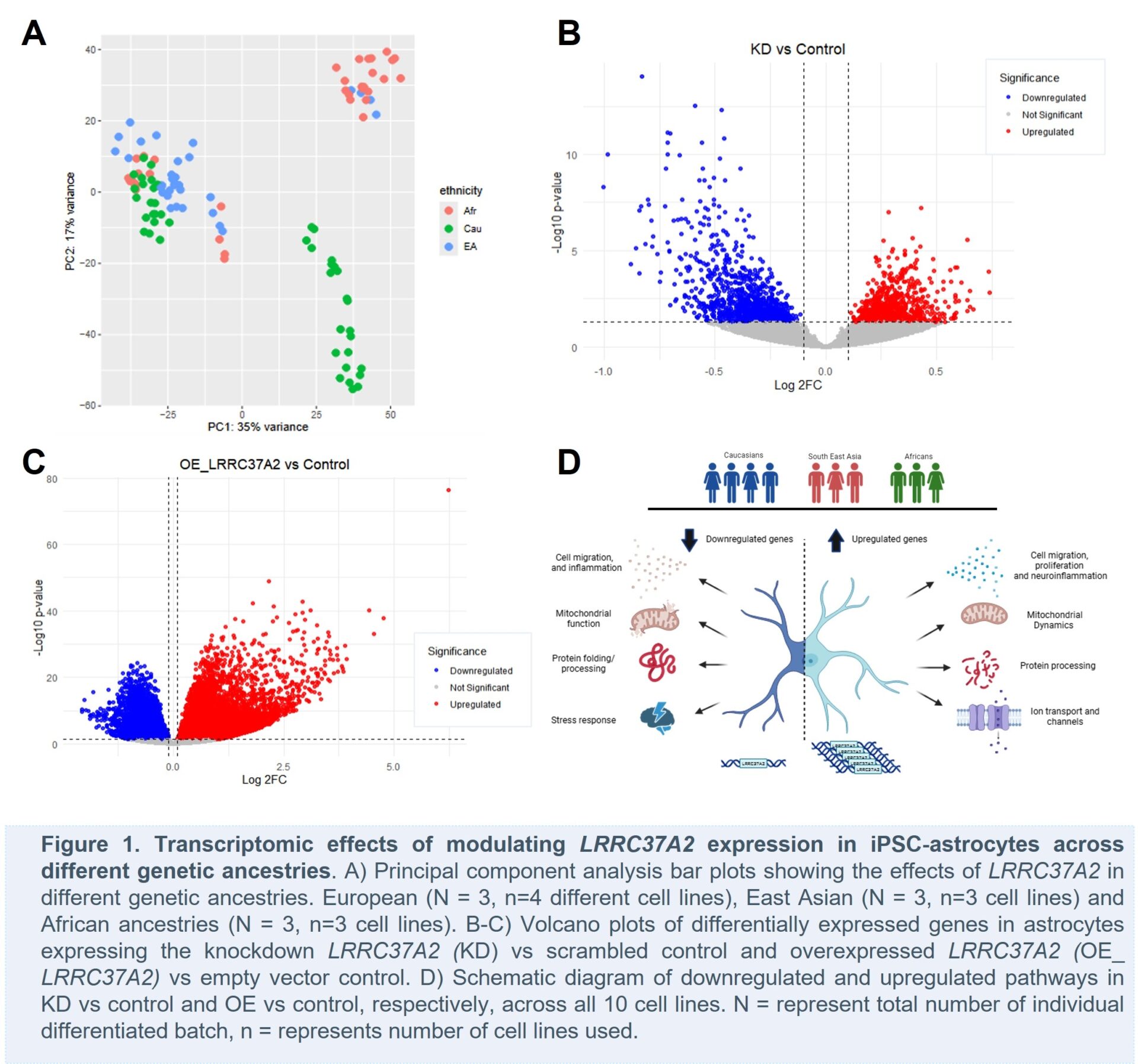
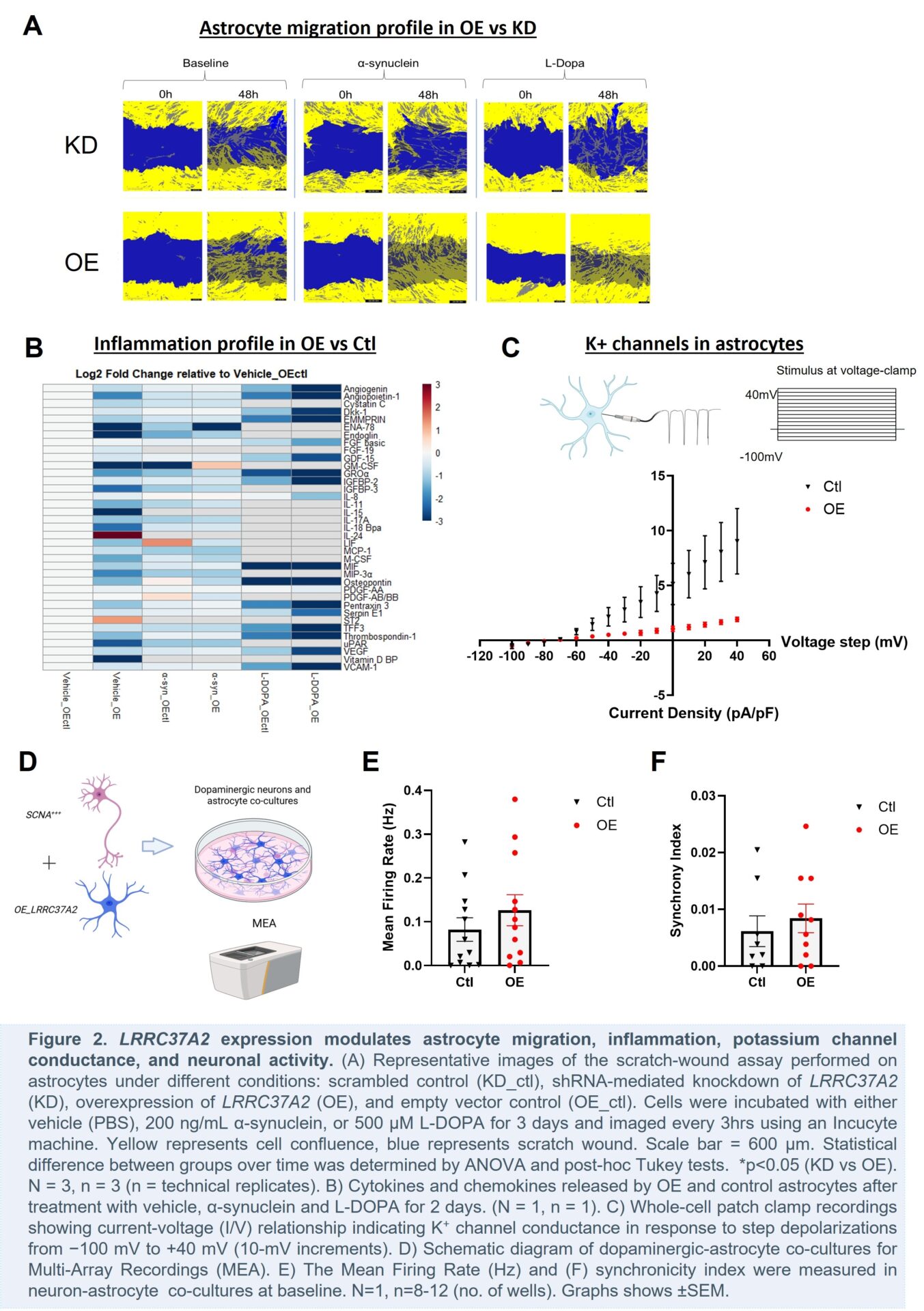
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disorder characterised by motor and non-motor symptoms. We have previously implicated the LRRC37A2 gene as a potential modifier of PD risk1; however, its cellular role remains poorly understood, partly due to its exclusion from genetic analyses. Located at the 17q21.31 ‘MAPT’ locus, a region prone to genomic instability, LRRC37A2 exhibits copy number variations (CNVs) that differ across major haplotypes, sub-haplotypes and genetic ancestry1,2,3. Elevated LRRC37A2 expression and CNVs are associated with reduced PD susceptibility1.
Objectives
This study aims to explore the role of LRRC37A2 in astrocyte function and its potential impact on neuronal health, further elucidating how LRRC37A2 confers protection from developing PD.
Methodology
Transcriptomic and functional analyses were used to assess cell- autonomous effects of modulating LRRC37A2 expression in induced pluripotent stem cell (iPSC)-derived astrocytes across European (n=4), East Asian (n=3), and African populations (n=3). To assess the non-cell autonomous effects of LRRC37A2, electrophysiological recordings were obtained from astrocytes mono-cultures and co-cultures of both astrocytes and dopaminergic neurons. Where appropriate, statistical difference between groups over time was determined by ANOVA and post-hoc Tukey tests.
Results
Transcriptomic profiling of astrocytes overexpressing LRRC37A2 across different genetic ancestries revealed upregulation of genes linked to cell migration, inflammation, mitochondrial dynamics, and autophagy lysosomal pathway (ALP) with no ancestry-specific differences in expression patterns. Functionally, we found that increased LRRC37A2 expression may exert a protective effect by modulating inflammatory signalling and enhancing astrocyte migration. Ongoing electrophysiological studies suggest that elevated LRRC37A2 expression in astrocytes also alters potassium channel conductance, leading to enhanced neuronal activity in co-cultured iPSC-dopaminergic neurons.
Conclusion
Collectively, these findings suggest that LRRC37A2 may play a protective role in PD by modulating astrocyte migration, inflammation, ion channel conductance, and neuronal activity. Further studies will examine the effects of astrocytic LRRC37A2 on mitochondrial dynamics and the ALP, as well as its non-cell autonomous effects on dopaminergic neurons carrying PD-related mutant genes. Elucidating the neuroprotective role of astrocytic LRRC37A2 and its therapeutic potential in treating PD may lead to the development of novel therapeutic strategies for this devastating disease.
Ethical Considerations
The iPSC lines used in this study were derived from donors who provided informed consent for the use of their cells in research. The iPSC lines were anonymized to protect donor privacy. The use of iPSCs in this study complies with the UK’s Human Tissue Act and the EU’s GDPR.