Introduction
Insulin resistance is a hallmark of type 2 diabetes, affecting over 4 million people in the UK1.While insulin signalling is classically associated with peripheral glucose homeostasis, insulin receptors are highly expressed in the brain, including in astrocytes, which integrate metabolic and hormonal cues to regulate whole-body homeostasis. Notably, short-term high-fat diet exposure induces mitochondrial fragmentation in astrocytes, which is sufficient to cause central insulin resistance2. However, the molecular pathways regulating astrocytic insulin sensitivity remain poorly defined.
Hedgehog signalling is best known for its role in development and oncogenesis but also regulates metabolism in peripheral tissues and the central nervous system. In addition to canonical Gli-mediated transcription, hedgehog signalling is involved in rapid non-canonical pathways. [BF1] Pharmacological hedgehog inhibitors enhance glucose uptake in mice3 and hedgehog signalling has been implicated in regulating mitochondrial dynamics in the brain4. Whether hedgehog signalling modulates insulin action in astrocytes remains unknown.
This study aimed to determine whether either canonical or non-canonical hedgehog signalling regulates astrocytic insulin sensitivity and mitochondrial dynamics.
Methods
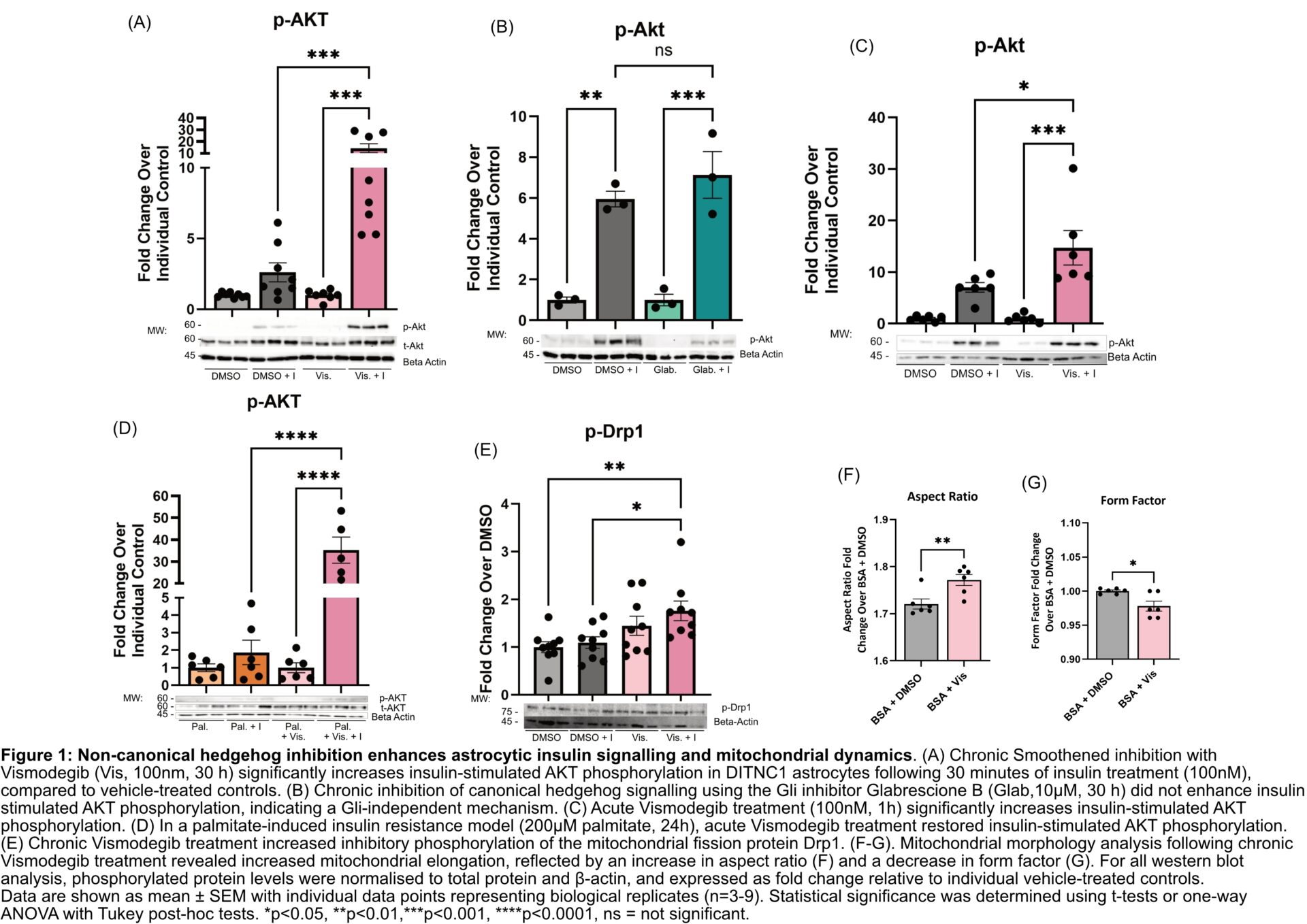
DITNC1 rat astrocytes were treated chronically (30 hours) with the Smoothened inhibitor Vismodegib (100 nM) or the Gli inhibitor Glabrescione B (10 μM), or acutely with Vismodegib (100 nM, 1 hour), followed by insulin stimulation (100 nM, 30 minutes) after serum starvation. Insulin resistance was induced using palmitate (200 μM, 24 hours) prior to acute Vismodegib treatment. Insulin signalling was assessed by western blotting for phosphorylated AKT and inhibitory phosphorylation of Dynamin-related protein 1 (Drp1). Mitochondrial morphology was quantified following chronic Vismodegib treatment using Mitotracker Red staining and confocal microscopy with Airyscan, assessing aspect ratio and form factor. Statistical analyses used t-tests or one-way ANOVAs with Tukey post-hoc tests (p<0.05).
Results
Chronic Smoothened inhibition with Vismodegib significantly enhanced insulin signalling, producing a 5.5-fold increase in insulin-stimulated AKT phosphorylation compared to the control, insulin treated, cells (p=0.0006, n=9, 1A). In contrast, chronic inhibition of canonical hedgehog signalling using the Gli inhibitor Glabrescione B failed to increase insulin-stimulated AKT phosphorylation (p=0.5633, n=3, 1B), indicating a non-canonical mechanism.
Consistent with this, acute Vismodegib treatment (1 hour), a duration insufficient to alter Gli1 expression as assessed by RT-PCR, significantly increased insulin-stimulated AKT phosphorylation (2-fold increase, p=0.0164, n=6, 1C) when compared with control-insulin treated cells. In a palmitate-induced insulin resistance model, acute Vismodegib robustly rescued insulin signalling, resulting in an 18.8-fold increase in insulin-stimulated AKT phosphorylation compared to palmitate-treated controls (p<0.0001, n=6, 1D).
Chronic Vismodegib treatment also increased inhibitory phosphorylation of the mitochondrial fission protein Drp1 (1.6-fold increase, p=0.0237, n=9, 1E). Mitochondrial morphology analysis revealed increased elongation, reflected by an increase in aspect ratio (3% increase, p=0.0095, n=6, 1F) and a reduction in form factor (2.21% decrease, p=0.0146, n=6, 1G).
Conclusion
Non-canonical hedgehog signalling enhances astrocytic insulin sensitivity and modulates mitochondrial dynamics independently of Gli-mediated transcription. Acute Smoothened inhibition is sufficient to rescue insulin resistance, while chronic modulation promotes mitochondrial elongation. Together, these findings position hedgehog signalling as a previously unrecognised regulator of astrocytic insulin action, with potential to improve insulin sensitivity in insulin-resistant brain states, including diabetes, obesity, and neurodegeneration.