Background:
Parkinson’s is a neurodegenerative disease characterised by the formation of α-synuclein aggregates and dopaminergic neuron loss in the substantia nigra, leading to debilitating movement symptoms (Morris et al, 2024). Chronic inflammation in the brain is present in a wide range of neurodegenerative diseases, including Parkinson’s. While research has traditionally focussed on the neurons affected by degeneration, our work explores the role of astrocytes, mediators of inflammation in the brain. In response to inflammation, astrocytes become “reactive”, switching from a neurosupportive to neurotoxic phenotype. Although animal models of Parkinson’s have shown that these reactive astrocytes cause neurodegeneration, the functional alterations elicited by inflammation that lead to neuronal loss are yet to be fully understood (Liddelow and Barres, 2017).
Results:
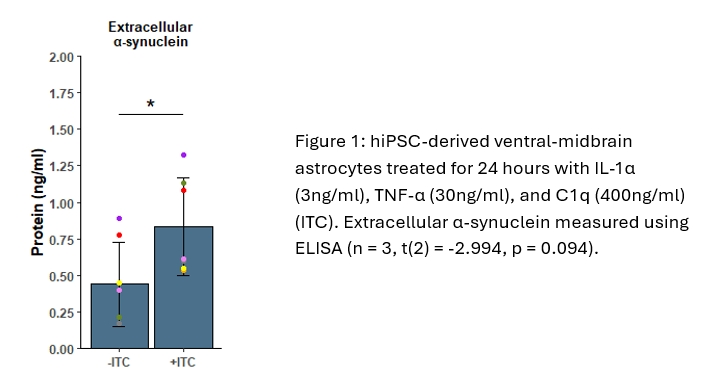
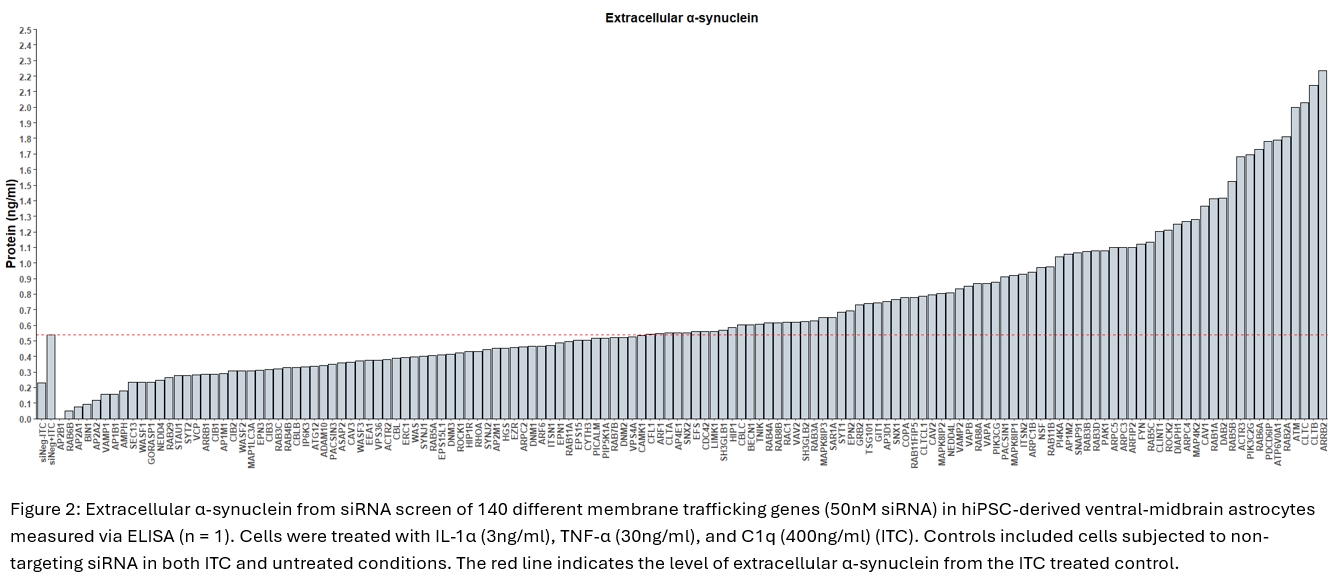
Using hiPSC-derived ventral-midbrain astrocytes (Crompton et al, 2021), we have identified a novel response in reactive astrocytes wherein vitro conditions recapitulating pro-inflammatory signals from activated microglia causes astrocytes to upregulate release of α-synuclein (figure 1). α-synuclein in the astrocyte conditioned media was measured by ELISA, and when comparing the untreated control (mean = 0.439 ng/ml, SD = 0.291 ng/ml) against the reactive astrocytes (mean = 0.835 ng/ml, SD = 0.333 ng/ml) there is a significant difference between the two conditions (n = 7, p = <0.05). To identify the mechanisms governing this response we have conducted a targeted siRNA screen of membrane trafficking genes which has revealed a number of exciting candidates including a component of the lysosomal V-ATPase (figure 2). To validate this we further targeted this complex with a shRNA and we found that the α-synuclein release associated with astrocyte reactivity was further increased (scramble shRNA mean = 1.475, SD= 0.257 vs V-ATPase shRNA ) mean = 2.582 ng/ml, SD = 0.477 ng/ml; two-way ANOVA with Tukey’s post-hoc test (figure 3, n = 3, p = <0.01).
Conclusions:
Ventral-midbrain astrocytes release α-synuclein when reactive in response to neuroinflammation contributing to pathology in a previously unidentified way. This suggests that astrocyte involvement in synucleinopathy may extend beyond clearance of neuronally secreted protein. This release of endogenous α-synuclein is upregulated by inhibition of proper lysosomal acidification, pointing to autolysosomal dysfunction as a possible causative mechanism behind this response.