Introduction
Astrocytes, a major glial cell type in the brain, play essential roles in maintaining neuronal function. In brain regions affected during neurodegenerative diseases such as Parkinson’s, reactive forms of these abundant cells are readily detected (1). Parkinson’s is characterised by the loss of dopaminergic neurons in the midbrain, a process associated with the intraneuronal formation of Lewy bodies and early inflammation. Lewy bodies are composed of misfolded α-synuclein aggregation, a protein central to synaptic neurotransmission and widely accepted as a diagnostic hallmark of Parkinson’s. Under physiological conditions, astrocytes help prevent buildup of extracellular α-synuclein through phagocytosis (2); however, their potential contributions to α-synuclein pathobiology and Parkinson’s progression are less well understood. Our mechanistic analysis of astrocytic α-synuclein handling suggests that during inflammation, reactive ventral midbrain astrocytes are primed to release α-synuclein into the extracellular environment. Additionally, lysosomal inhibition potentiates astrocytic α-synuclein release, suggesting that mishandling in the astrocyte endolysosomal/autolysosomal compartments might contribute to Parkinson’s-associated Lewy body pathology.
Methods
Ventral-midbrain astrocytes (vmAstros) (3) were differentiated from normal patient-derived iPSCs and used to measure intra- and extracellular α-synuclein levels with or without inflammatory stimulation (IL-1α(3ng/ml); TNF-α (30ng/ml); C1q (400ng/ml)). Based on candidate genes identified in an siRNA membrane trafficking library was used to screen for pathways involved in α-synuclein release (ELISA), tandem shRNAs/rescue lentiviral vectors and lysosomal inhibitors (BafA1; pepstatin A, E-64) were used tobegin assess lysosomal involvement and links to canonical and non-canonical autophagy pathways. For these assays, immunoblotting and ELISA-based analysis were used to measure intracellular and secreted α-synuclein, respectively.
Results
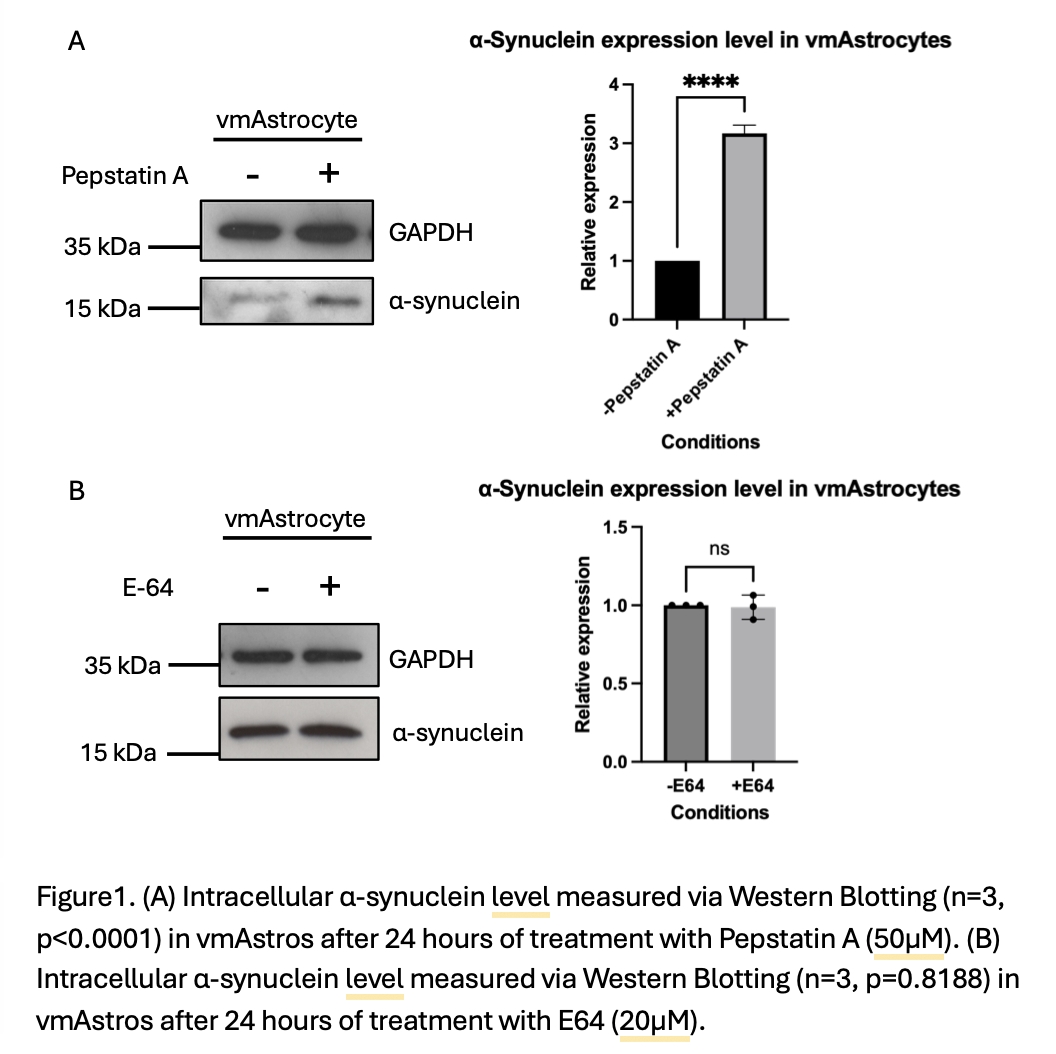
Using the library of siRNAs targeting membrane trafficking genes with ELISA-based analysis of α-synuclein release form reactive vmAstros, we found evidence of involvement of the lysosomal system in α-synuclein release. This has been validated using shRNA against selected components of the lysosomal V-ATPase. Focusing on α-synuclein turnover, treatment with pepstatin A (24hrs), vmAstros showed significantly higher (3.2-fold; SE<0.08) α-synuclein levels than in quiescent vmAstros (Fig. 1A; unpaired t-test, p<0.0001). By contrast, E-64 treatment (24hrs) did not alter endogenous α-synuclein levels (Fig. 1B; unpaired t-test, p=0.8188). We are continuing to examine the involvement of the endolysosomal and autophagy systems in pathological vmAstro α-synuclein handling in Parkinson’s.
Conclusion
A defective endolysosomal system is implicated in pathological α-synuclein release from reactive vmAstros. This could establish a positive feedback loop of inflammation and a-synuclein release in Parkinson’s. Given the increasing well-recognised links between endolysosomal dysfunction and Parkinson’s susceptibility, our work to determine how this pathway is controlled at a mechanistic level is of key value.