Introduction:
Human genetics have revealed the importance of microglia in Alzheimer’s disease (AD). Recent work has shown that the microglial response to amyloid pathology dictates the transition to Tau pathology, or not. Human genetics for AD has currently identified 75+ risk genes. However, how these risk genes and new genes combine in pathways to coordinate microglial function and how these processes contribute to Tau pathology and resilience to AD are not well understood.
Aim/Objective:
The objective of our studies is to gain new insights into the biological processes which underlie AD and longevity (or resilience to AD) using an unbiased approach to integrate common human genetic variation of human lifespan and AD from GWAS.
Methods:
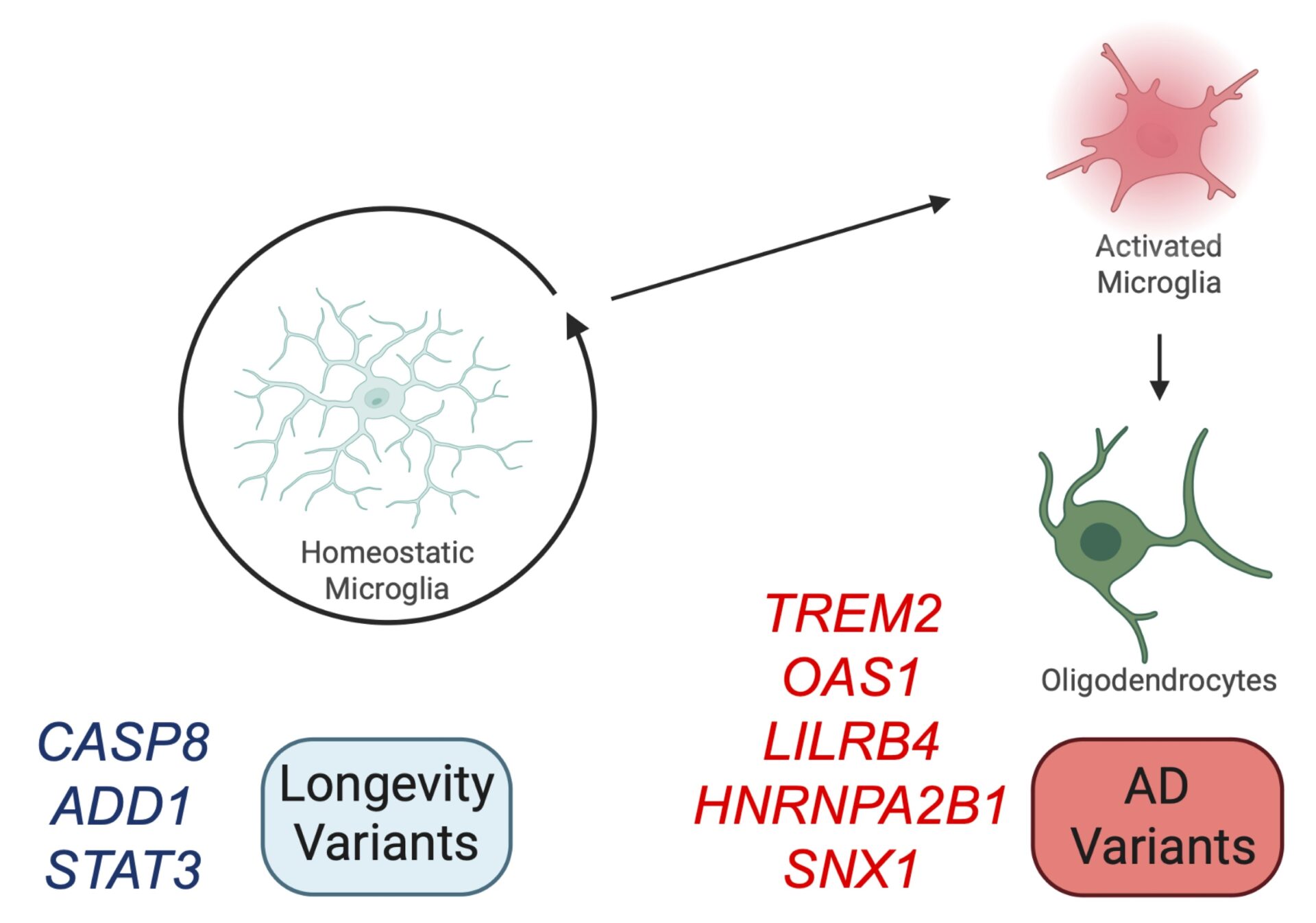
AD GWAS summary statistics data from the IGAP stage 1 GWAS meta-analysis of 21,982 AD cases and 41,944 cognitively normal controls (Kunkle et al. 2019). Three longevity GWASs covering healthspan (300,477 individuals), parental lifespan (1,012,240 parents) and longevity (defined by 11,262 individuals surviving to the 90th percentile of life and 25,483 controls whose age at death corresponds to the 60th survival percentile; Timmers et al. 2020). Bulk hippocampal RNA-sequencing (RNA-seq) datasets were generated from wild-type C57BL/6J mice, amyloid mice (APPK670N/M671L and PSEN M146V mutant lines) and tau mice (MAPT P301L) of ages 2-, 4-, 8- and 18-months-old (n = 2-7 mice per genotype per age group; data available from Mouseac.org; Salih et al. 2019), and non-diseased human hippocampi (from 196 individuals from the Genotype-Tissue Expression (GTEx) Project v8; gtexportal.org/home/datasets; GTEx et al. 2015). Single-cell hippocampal RNA-seq of microglia isolated from wild-type mice of ages 3-, 6-, 12- and 21-months-old (n = 2 mice per sex/age, pooled; Sala Frigerio et al. 2020). A two-step approach was used to identify biological pathways dysregulated with age and that are enriched for genes associated with AD risk or longevity. First, common human genetic variation associated with AD or ageing at the gene-based level were identified by calculating the joint summary of all SNPs in the gene. Second, assessment of statistical enrichment of associated AD and longevity genes in age- and AD-related gene co-expression networks, generated from the transcriptomic profile of the human and mouse hippocampus (Graham et al. 2025).
Results:
Our work reveals that genetic variation associated with AD was enriched in both human microglia (P = 1e-5, enrichment bootstrap-based test) and oligodendrocytes (P = 0.033, enrichment bootstrap-based test), which show the strongest increases in gene expression with ageing, and contribute to Tau pathology. Whereas, longevity-associated genetic variation was enriched in homeostatic microglia (P = 0.032, enrichment bootstrap-based test), which may drive “inflammageing.” Thus, we observe that variants contributing to resilience, AD and Tau pathology balance different aspects of microglial function.
Conclusions:
Our findings have important implications for developing markers indicating the physiological age of the brain and Tau pathology, alongside better diagnosis of disease and new targets for therapeutic intervention.