Physiology News Magazine
A chronology of chronobiology
From plants in cupboards and bovine pineal "popcorn" to mood disorder and cancer therapy
Features
A chronology of chronobiology
From plants in cupboards and bovine pineal "popcorn" to mood disorder and cancer therapy
Features
Thomas C Erren, Institute and Policlinic for Occupational Medicine, Environmental Medicine and Prevention Research, University Hospital of Cologne, Germany
https://doi.org/10.36866/pn.113.32
Life on Earth evolved under cyclic light-dark conditions due to our planet’s rotation. Circadian physiology, facilitating interplays between light and time and life, allowed species to anticipate daily environmental changes and align their internal time accordingly for competitive edges in survival and evolution. The causal web connecting environmental and circadian times includes complex interactions between: (1) light; (2) our eyes as dual sense organs; (3) the suprachiasmatic nuclei (SCN) – or master clock – in the anterior hypothalamus; (4) peripheral clocks; and (5) melatonin molecules secreted from the pineal gland in response to environmental time cues. Clockwork facets discovered over some 300 years shed light on the evolutionary history of species on Earth, including humans. Importantly, research into determinants and effects of the temporal organisation of physiology can offer fascinating insights into this timing network with possible high relevance for individual and public health.
Indeed, we all have an internal timing system that organises our physiology over time in alignment with periodic zeitgebers (from German, “time-giver”) such as the light-dark cycle. For young(er) readers, what challenges or perspectives may derive from grappling with the concept of internal time? First, familiarise yourself with roots of chronobiological thinking. Pause here: consideration of an evolutionary legacy of internal time and circadian organisation across species, including humans, must stimulate further thoughts on integrated circadian physiology. Second, begin to answer how – at an evolutionary scale – abrupt changes such as making the night into day by widely available man-made light since the late 19th century may disrupt ancient health-promoting systems. Third, understanding and possibly clarifying terminology can be an excellent start for chronobiological considerations such as how health may be affected by conflicts between internal time and, for instance, work at chronobiologically unanticipated times.
1720s – 1930s: The foundation of the field
To begin, four key studies can be noted as providing the platform for significant advancements in the field. First and foremost, de Mairan’s observation regarding the Mimosa pudica plant marks the dawn of chronobiology in 1729. Until his “experiment in a cupboard”, the assumption was that the plant opened in the day due to sunlight and closed at night due to darkness. Yet, when de Mairan exposed the plant to total darkness, opening and closing of the flowers continued. His discovery was that an intrinsic timekeeper must be present which is responsive to both light/dark and non-light/dark influences. From here, it took more than 200 years before Kleitman and Richardson entered Mammoth Cave, a location devoid of natural light, to investigate intrinsic timekeepers in humans (Fig. 1). Assuming no environmental time cues, they attempted to switch to a 28-hour sleep-wake cycle. Despite lasting 32 days in the cave, their results of n =2 were inconclusive. Nonetheless, this early attempt to determine whether humans have endogenous clocks, was pioneering for the field. In addition, two decades (1917) before the Mammoth Cave expedition, one peculiar observation prepared the ground for identifying melatonin – now considered one of the body’s key time messengers. Namely, when McCord and Allen fed bovine pineal gland materials to Rana pipiens tadpoles, a dose-dependent skin lightening occurred and larger viscera could be seen. When investigating vitiligo-like disruptions in skin pigmentation in the 1950s, Lerner et al. followed up on these observations to discover that a pineal indole (melatonin) caused melanin granules in frog melanocytes to aggregate and their skin to lighten.

1950s – 2000s: Terminological support for the field
Unambiguous terminology to describe falsifiable concepts is a conditio sine qua non for interpretable research. In the 1950s, there was a surge in chronobiological research, and terminology was crucial. In 1951, the German chronobiologist Aschoff termed external or environmental time cues “zeitgeber”. These environmental factors were named “synchronisers” by Halberg et al. and “entraining agents” by Pittendrigh and Bruce but the three terms are synonymous. In 1958, Lerner et al. called the pineal gland factor “melatonin” after showing it inhibited melanocyte-stimulating hormone (MSH) and lightened skin colour in frogs. In 1959, Halberg, after discussion with McDonald (and others) of the Department of Classics at the University of Minnesota, proposed the term “circadian” – deriving from Latin: “circa” (about) and “dies” (day). In 1974, Ehret coined “chronotype” as the temporal phenotype of an organism which captures a biological trait regarding when individuals tend to be awake or asleep. While “circadian disruption” is not clearly defined, “chronodisruption” as the split physiological nexus of internal [circadian clock] and external times was proposed in 2003. Already in 1959, Halberg emphasised what is still true today: “One of the difficulties in correlating the various views and opinions on how periodic biological phenomena are established and maintained lies in the confusion of terms used” (Halberg et al., 1959). In this regard, the 1960 Cold Spring Harbor Symposium: Biological Clocks, Vol. XXV, proved a landmark event, bringing together nestors of chronobiology, laying down “laws” for future investigations, and providing stimulating seeds and feeds for the nascent field.
1950s – 2000s: Nobel-worthy discoveries
Paved by the earlier work, the 1950s brought about a surge in chronobiological research. The selection for this section starts with Axelrod, the 1970 Nobel laureate who completed his PhD at the “early age” of 43. Part of the work which won him the Nobel, viz “Noradrenaline: fate and control of its biosynthesis”, involved investigating the pineal gland which produces the bulk of melatonin that enters the blood. Axelrod clarified relationships between melatonin, tryptophan, and serotonin, which all follow circadian rhythm. The isolation of melatonin is a discovery saga of its own. It took Lerner et al. no less than 250,000 bovine pineal glands (Fig. 2 – looks like a giant bag of popcorn!) to ultimately characterise the “Dracula molecule” or “chemical expression of darkness”. Today we know that melatonin rhythms serve both as a clock and calendar (Reiter, 1993). In the 1960s, Aschoff et al. followed up the landmark experiment by Kleitman and Richardson with observation of humans in underground bunkers. Ultimately, it was concluded that human volunteers had endogenously generated circadian sleep-wake cycles.
One new era for research into chronobiology came with the discovery of first clock mutants in Drosophila melanogaster (fruit fly) in 1971 and in Mesocricetus auratus (Syrian or golden hamster) in 1988, two examples of (if you will) “natural experiments” which were profitably exploited (Konopka & Benzer, 1971; Ralph & Menaker, 1988). The former led to work which won Hall, Rosbash and Young last year’s Nobel award for discoveries of molecular mechanisms controlling the circadian rhythm. Regarding the latter, transplantation of tau mutant SCN into SCN-ablated wild-type animals inflicted the donor’s rhythm on the host; namely, they gained the homozygous mutant’s 20 hours sleep-wake cycle instead of wild-type 24 hours. This confirmed the SCN (Fig. 3) as the site of the master circadian pacemaker (Ralph et al., 1990).
A new era for photoreception research arrived with understanding that the eye is a dual sense organ. In 1991, circadian responses to light were observed in mice with hereditary retinal disorders despite loss of image-forming visual photoreceptors by Foster et al. Vertebrate ancient (VA) opsin discovery in salmon was an intermediate step to ultimately demonstrate that intrinsically photosensitive retinal ganglion cells (ipRGCs) contain the photopigment melanopsin and contribute critically to circadian (non-image-forming) vision (Soni et al., 1998). In 2007, non-rod, non-cone photoreceptors were demonstrated in two humans with rare eye diseases. They had no rods or cones but intact ipRGCs and photoentrainment proved possible (Zaidi et al., 2007). Hattar et al. (2002, 2006) illustrated “hardware” connections of ipRGCS to the SCN and further architecture and projections throughout the brain. These await linkage to functional questions: How is circadian time relayed throughout the body? How are health and disease linked with circadian organisation? How can we translate chronobiological insights to combat disease?
Suffice to say, there are numerous discoveries which did not make the cut for this article. For those interested in more, the 1960 Cold Spring Harbor Symposium is an excellent place to start.

1980s-present: Time for translation
First answers to the latter question were provided by Rosenthal et al., (1984). The sleep researchers proposed seasonal affective disorder (SAD) as a syndrome occurring at regular times of the year. On the basis of 29 patients, they concluded that most SAD individuals had a bipolar affective disorder, appeared responsive to changes in latitude-associated photoperiod, and exhibited impaired sleep. Preliminary exploration of how bright light could have antidepressant effects was the prelude to further developments in light therapy.
Another example of translating chronobiological insights into clinical practice is that the value, efficacy, and safety of pharmacological treatments should be tailored to individual internal time. Rather than treating physiology and pathophysiology as constants where one medication timing fits all patients’ circadian times, chronobiology suggests that circadian organisation varies over time and across individuals. Applying this to disease, Lévi and colleagues have been exploring how cancer treatments can be improved by considering circadian timing (Martineau-Pivoteau et al., 1996).
Future challenges and perspectives
Studying effects of man-made light pollution on health and disease in organisms, including humans, is a future challenge. For instance, in 2007, the International Agency for Research on Cancer concluded that shift-work involving circadian disruption is a probable human carcinogen (Group 2A). A major problem, however, is that the key link in the plausible chain of cancer causation, viz circadian disruption, has neither been defined nor were suggestions made on how to assess doses of circadian disruption. Various metrics to allow computation of chronodisruption or circadian misalignment or sleep deficiency all build upon individuals’ discrepant internal and external times due to e.g. shift-work. But there is a big “if” regarding all these metrics; namely, how chronotype or internal time can be appropriately assessed over prolonged periods (months/years) remains unanswered (Erren et al., 2018).
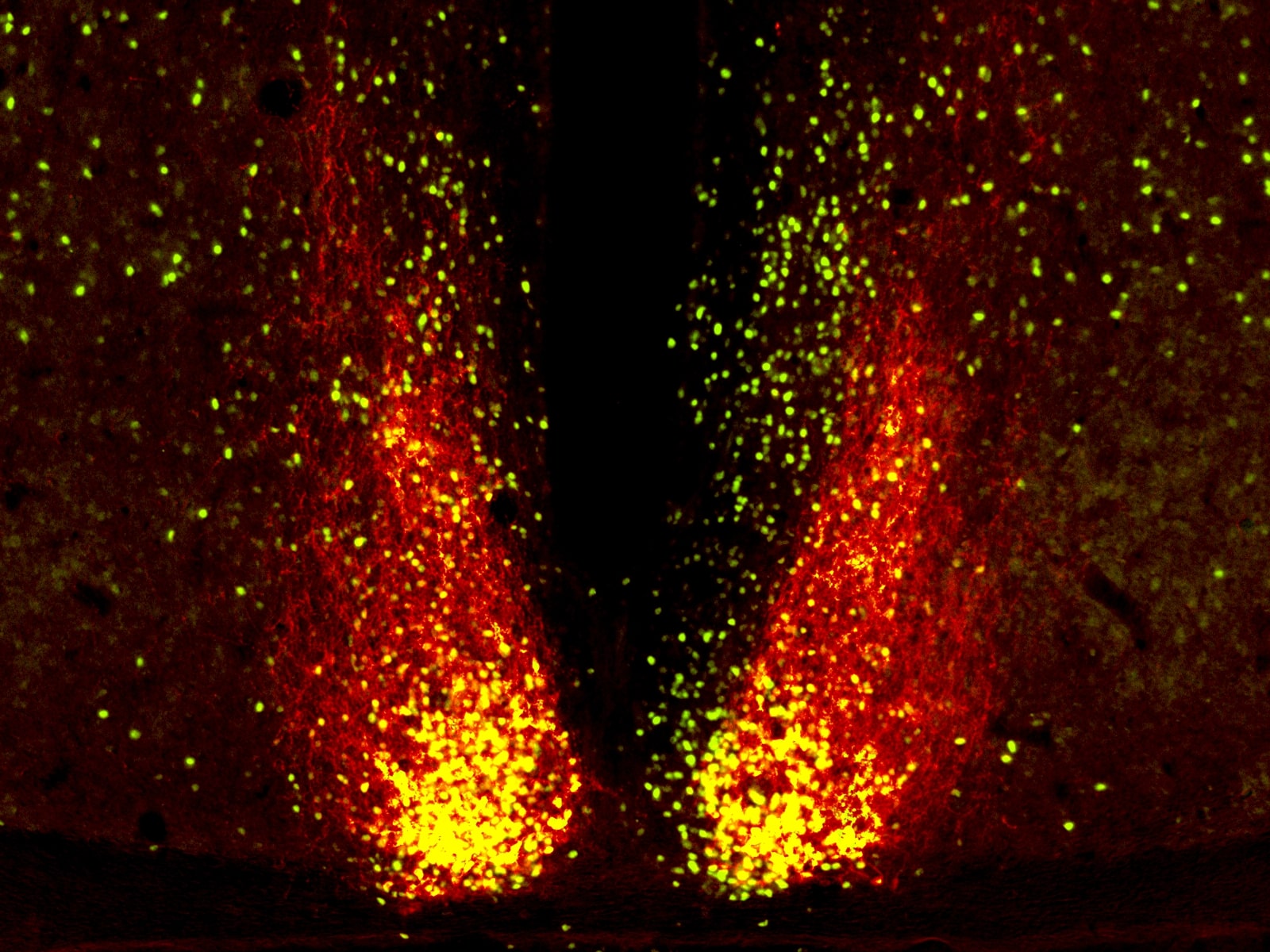
It is also of interest whether individuals vary in their susceptibility to circadian challenges. In this vein, researchers have begun to explore whether Perinatal Light Imprinting of Circadian Clocks and Systems (PLICCS) renders individuals born under winter light conditions or photoperiods more susceptible to circadian challenges later in life than those born under summer light conditions (Lewis & Erren, 2017).
Overall, from a public health point of view, a promising approach to improve circadian-related health and performance may be to strive for zeitgeber hygiene. That is to say, strive to align diverse zeitgebers such as light, food, activities, and social factors such that they interact synergistically rather than antagonistically. A case in point to fuel readers’ curiosity and scientific creativity can be recent observations by Wehr (2018).
His report of potential synchronicity of mood cycles in rapid cycling bipolar disorder patients with lunar cycles, if not falsified, could impact how we understand disease causation, diagnosis and prevention and, more generally, chronobiology. If true, bipolar patients might switch rapidly between two rhythmic periods: the circadian period seemingly free-running, viz close to 25 hours, when symptomatic and 24 hours (entrained to natural light-dark cycle) when not. Now, if relevant moon-associated zeitgeber signalling was corroborated, what we call endogenous circadian periods today might be considered a misnomer because what we measured might have been affected by un(der)-appreciated zeitgeber information from the Moon.
In conclusion, careful and open-minded chronobiology-based and chronobiology-targeted research can pave the road to important insights into circadian time, on the one hand, and health and disease, on the other.
References
Erren TC, Groß JV, Lewis P (2018). Computing sleep deficiency. Journal of Sleep Research 27, e12630
Halberg F, Halberg E, Barnum CP et al. (1959). Physiologic 24-hour periodicity in human beings and mice, the lighting regimen and daily routine. In Photoperiodism and Related Phenomena in Plants and Animals. American Association for the Advancement of Science 55, 803-878.
Hattar S, Liao HW, Takao M et al. (2002). Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295, 1065-1070.
Hattar S, Kumar M, Park A et al. (2006). Central projections of melanopsin-expressing retinal ganglion cells in the mouse. Journal of Comparative Neurology 497, 326-349.
Konopka RJ, Benzer S (1971). Clock mutants of Drosophila melanogaster. Proceedings of the National Academy of the United States of America 68,
2112-2116.
Lewis P, Erren TC (2017). Perinatal light imprinting of circadian clocks and systems (PLICCS): The PLICCS and cancer hypothesis. Frontiers in Oncology 7, 44.
Martineau-Pivoteau N, Levi F, Rolhion C et al. (1996). Circadian rhythm in toxic effects of cystemustine in mice: relevance for chronomodulated delivery. International Journal of Cancer 68, 669-674.
Ralph MR, Menaker M (1988). A mutation of the circadian system in golden hamsters. Science 241, 1225-1227.
Ralph MR, Foster RG, Davis FC et al. (1990). Transplanted suprachiasmatic nucleus determines circadian period. Science 247, 975-978.
Reiter RJ (1993). The melatonin rhythm: both a clock and a calendar. Experientia. 49, 654-664.
Rosenthal NE, Sack DA, Gillin JC et al. (1984). Seasonal affective disorder. A description of the syndrome and preliminary findings with light therapy. Archives of General Psychiatry 41, 72-80.
Soni BG, Philp AR, Foster RG et al. (1998). Novel retinal photoreceptors. Nature 394, 27-28.
Wehr TA (2018). Bipolar mood cycles and lunar tidal cycles. Molecular Psychiatry 23, 923-931.
Zaidi FH, Hull JT, Peirson SN et al. (2007). Short-wavelength light sensitivity of circadian, pupillary, and visual awareness in humans lacking an outer retina. Current Biology 17, 2122-2128.
