
Mitochondria and sleep-control neurons
Raffaele Sarnataro & Cecilia D. Velasco
Centre for Neural Circuits and Behaviour, University of Oxford, Oxford, United Kingdom
Mitochondria play a pivotal role in regulating one of the most essential and restorative physiological states: sleep. Raffaele Sarnataro & Cecilia D. Velasco tell us more.
Physiology cannot be understood without the concept of homeostasis. Defined as the self-regulating process that maintains internal stability, homeostasis enables the body to adapt to a constantly changing environment that may disrupt equilibrium.
Homeostatic control of internal states is primarily coordinated by the brain, which acts as a control centre that monitors and responds to signals from across the body. For example, whole-body energy homeostasis is regulated by the central nervous system, which balances food intake with energy expenditure (Roh et al., 2016). Mitochondria, the cellular metabolic hubs, play a key role in this process by dynamically adapting their morphology and consequently their function, in response to changes in energy availability and demand (Haigh et al., 2020). Elongated and fused mitochondria are energetically more efficient than small and fragmented (or fissured) ones (Hoitzing et al., 2015).
These adaptive changes illustrate how cells finely tune their metabolism to match fluctuating energy demands. However, the influence of mitochondria extends far beyond the nutritional state of the body. They also play a pivotal role in regulating one of the most essential and restorative physiological states: sleep (Sarnataro et al., 2025).
Sleep and wakefulness
The alternation between sleep and wakefulness is homeostatically controlled: the longer an animal remains awake, the greater its need for sleep becomes. In the fly, sleep-control neurons projecting to the dorsal fan-shaped body (dFBNs, Figure 1) monitor sleep need and promote sleep (Donlea et al., 2011; Hasenhuetl et al., 2024; Kempf et al., 2019; Sarnataro et al., 2025). In a recent study, researchers performed single-cell transcriptomics comparing rested and sleep-deprived flies and found that, in dFBNs, but not throughout the brain, sleep deprivation triggers a prominent upregulation of genes involved in metabolism and ATP synthesis (Sarnataro et al., 2025).
Loss of sleep elevates ATP levels in dFBNs, leading to electron accumulation in the respiratory chain and the formation of reactive oxygen species, which signal rising sleep need (Kempf et al., 2019; Sarnataro et al., 2025). Structurally, mitochondria in these sleep-control neurons respond dynamically to sleep need. After sleep deprivation, they become fragmented, display enhanced mitophagy, and increase the number of contact sites with the endoplasmic reticulum. These morphological changes reverse after sleep recovery, with mitochondria becoming more elongated (Sarnataro et al., 2025).
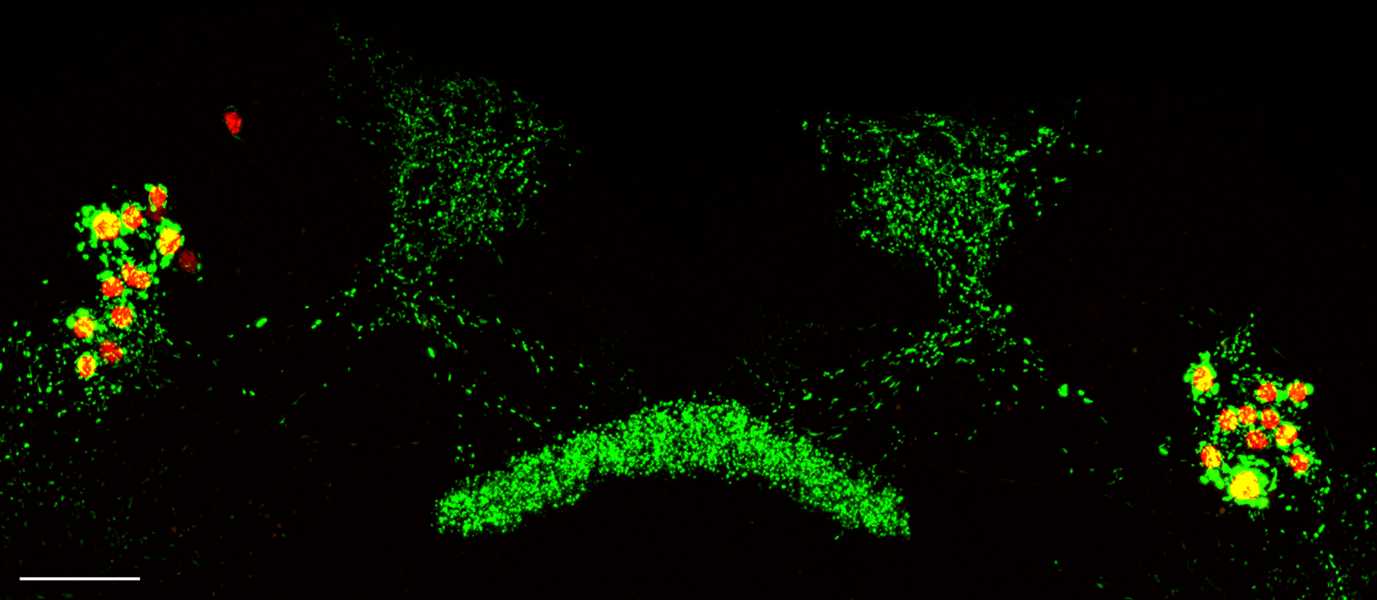
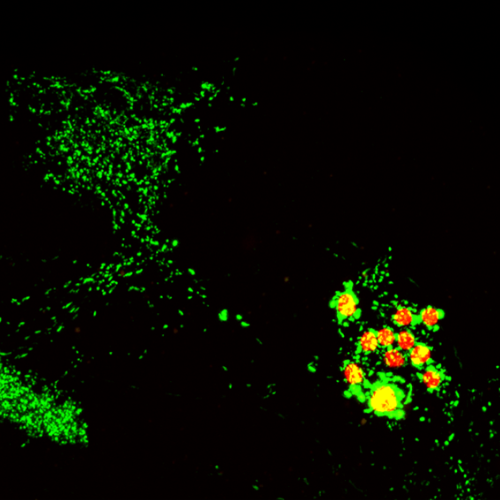
Dorsal fan-shaped body neurons (dFBNs), labelled by R23E10-GAL4, expressing a mitochondrially-targeted GFP (green) and the cell nuclei marker RedStinger (red). Cellular processes departing from the somata arborise in two hemispheric dendritic tufts and converge centrally to innervate the dorsal layers of the fan-shaped body. Scale bar = 25 μm
Altering sleep behaviour
Critically, manipulating mitochondrial morphology alters sleep behaviour: promoting fusion increases both neuronal excitability and sleep, whereas promoting fission has the opposite effect. While rearrangements in mitochondrial shapes by loss of sleep occur in various brain regions of several species (Sarnataro, 2025), in sleep-control neurons, they actively encode homeostatic sleep need rather than merely reflecting it. These findings parallel mechanisms described in feeding regulation (Dietrich et al., 2013).
This recent work, situated at the interface of metabolism and neuroscience, reveals that homeostatic control of sleep emerges from brain energy balance. This balance is sensed through a metabolic feedback loop embedded within the cellular powerhouses of neurons.
However, mitochondria are central not only to ATP production but also to calcium buffering (Cartes-Saavedra et al., 2025) and redox balance (Shadel and Horvath, 2015), which are critical processes for neuronal physiology. When these functions falter, the frail neuronal orchestration of mental processes becomes particularly vulnerable. Consequently, even subtle impairments in mitochondrial function can have disproportionate effects on brain health.
Faulty machinery
Disruptions in the machinery that governs mitochondrial fission and fusion are frequently associated with a range of neurodegenerative conditions (Alexander et al., 2000; Pickrell and Youle, 2015; Züchner et al., 2004). Elevated mitochondrial DNA content and heteroplasmy are molecular markers of major depressive disorder (Cai, Chang, et al., 2015; Cai, Li, et al., 2015), and altered mitophagy has been detected in psychotic patients (Khadimallah et al., 2022).
In many of these disorders, sleep disturbances are prevalent (Anghel et al., 2023). Although it remains difficult to determine whether such disruptions are a cause or a consequence of neuropathology. Mounting evidence indicates that they actively worsen disease progression in a vicious, self-reinforcing cycle (Anghel et al., 2023; Harris et al., 2021; Simmonds et al., 2025). The described recent work introduces a new mechanistic perspective on the metabolism–sleep axis (Sarnataro et al., 2025), suggesting that metabolic interventions could provide a means to break the bidirectional interplay between altered sleep and brain pathology.
References
Alexander C et al. (2000) OPA1, encoding a dynamin-related GTPase, is mutated in autosomal dominant optic atrophy linked to chromosome 3q28. Nature Genetics 26(2): 211–215. doi: 10.1038/79944
Anghel L et al. (2023) Sleep Disorders Associated with Neurodegenerative Diseases. Diagnostics 13(18): 2898. doi: 10.3390/diagnostics13182898
Cai N, Li Y et al. (2015) Genetic Control over mtDNA and Its Relationship to Major Depressive Disorder. Current Biology 25(24): 3170–3177. doi: 10.1016/j.cub.2015.10.065
Cai N, Chang S et al. (2015) Molecular Signatures of Major Depression. Current Biology 25(9): 1146–1156. doi: 10.1016/j.cub.2015.03.008
Cartes-Saavedra B et al. (2025) The roles of mitochondria in global and local intracellular calcium signalling. Nature Reviews Molecular Cell Biology 26(6): 456–475. doi: 10.1038/s41580-024-00820-1
Dietrich MO et al. (2013) Mitochondrial Dynamics Controlled by Mitofusins Regulate Agrp Neuronal Activity and Diet-Induced Obesity. Cell 155(1): 188–199. doi: 10.1016/j.cell.2013.09.004.
Donlea JM et al. (2011) Inducing sleep by remote control facilitates memory consolidation in Drosophila. Science 332(6037): 1571–1576. doi: 10.1126/science.1202249
Haigh JL et al. (2020) Mitochondrial Dynamics in the Brain Are Associated With Feeding, Glucose Homeostasis, and Whole-Body Metabolism. Frontiers in Endocrinology 11: 580879. doi: 10.3389/fendo.2020.580879
Harris, SS et al (2021) The Reciprocal Interaction Between Sleep and Alzheimer’s Disease. In: Engmann, O., Brancaccio, M. (eds) Circadian Clock in Brain Health and Disease. Advances in Experimental Medicine and Biology, vol 1344. Springer, Cham. doi: 10.1007/978-3-030-81147-1_10
Hasenhuetl PS et al. (2024) A half-center oscillator encodes sleep pressure. BioRxiv. doi: 10.1101/2024.02.23.581780
Hoitzing H et al. (2015) What is the function of mitochondrial networks? A theoretical assessment of hypotheses and proposal for future research. BioEssays 37(6): 687–700. doi: 10.1002/bies.201400188
Kempf A et al. (2019) A potassium channel β-subunit couples mitochondrial electron transport to sleep. Nature 568(7751): 230–234. doi: 10.1038/s41586-019-1034-5
Khadimallah I et al. (2022) Mitochondrial, exosomal miR137-COX6A2 and gamma synchrony as biomarkers of parvalbumin interneurons, psychopathology, and neurocognition in schizophrenia. Molecular Psychiatry 27(2): 1192–1204. doi: 10.1038/s41380-021-01313-9
Pickrell AM and Youle RJ (2015) The Roles of PINK1, Parkin, and Mitochondrial Fidelity in Parkinson’s Disease. Neuron 85(2): 257–273. doi: 10.1016/j.neuron.2014.12.007.
Roh E et al (2016) Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism. Experimental & Molecular Medicine 48(3): e216–e216. doi: 10.1038/emm.2016.4
Sarnataro R (2025) Neurobiology of mitochondrial dynamics in sleep. The Journal of Physiology. doi: 10.1113/JP288054
Sarnataro R et al. (2025) Mitochondrial origins of the pressure to sleep. Nature 645(8081): 722–728. doi: 10.1038/s41586-025-09261-y
Shadel GS and Horvath TL (2015) Mitochondrial ROS Signaling in Organismal Homeostasis. Cell 163(3): 560–569. doi: 10.1016/j.cell.2015.10.001
Simmonds E et al. (2025) Sleep disturbances as risk factors for neurodegeneration later in life. npj Dementia 1(1): 6. doi: 10.1038/s44400-025-00008-0
Züchner S et al. (2004) Mutations in the mitochondrial GTPase mitofusin 2 cause Charcot-Marie-Tooth neuropathy type 2A. Nature Genetics 36(5): 449–451. doi: 10.1038/ng1341



